
May take a minute to load the many images!
Organic Evolution in terms of the Implicate and Explicate Orders.
Part LXXV
Diptera (midges, mosquitoes, flies) (VII)
The evolutionary diversification in the Order Diptera revisited.
Lifting not-costalized (musidoroid) functional wing-type
(Descriptions taken from ROHDENDORF, 1951, with additions and additional Figures)
Representatives of the type
Peculiar wings of the lifting not-costalized (musidoroid) type are possessed by species of the family Musidoridae and Streblidae. See next Figure.
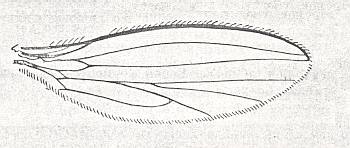
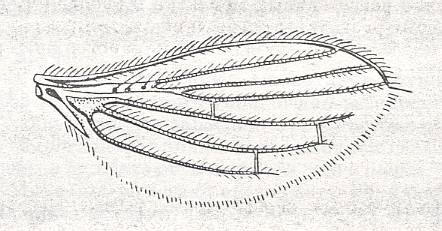
Figure 1 : Wings of representatives of the lifting not-costalized (musidoroid) type.
Top image : Musidora lutea Panz., Musidoridae. Female. Length 3.4 mm. (After Rohdendorf, 1951).
Bottom image : Pseudostrebla ribeiroi, Streblidae. Length 2 mm. (After Jobling).
(From ROHDENDORF, 1951)
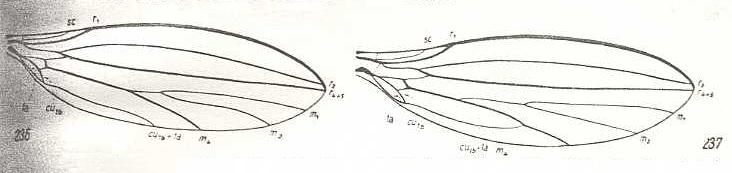
Figure 2 : Wing-venation of representatives of the lifting not-costalized (musidoroid) type.
Left image : Musidora strobli de Meij., Musidoridae. Male.
Right image : Musidora strobli de Meij., Musidoridae. Female.
(After HENNIG, 1954)
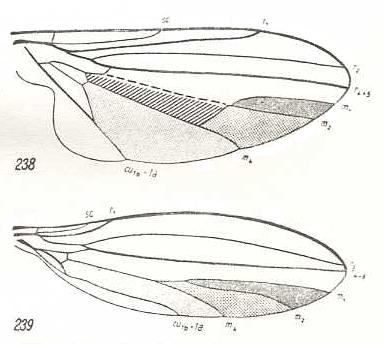
Figure 3 : Schemata, clarifying the wing-venation in Musidoridae.
The original condition (top image) approximately corresponds to the venation in Platypezina connexa Wahlb. (Clythiidae).
(After HENNIG, 1954)

Figure 4 : Lonchoptera lutea Panz., Lonchopteridae (= Musidoridae). Female. Length of insect about 4. mm.
(After CHINERY, in Elseviers insektengids voor West-Europa, 1983)
Size of wings
Absolute size of the wings small, varying from 1.5 (some Streblidae) to 4 mm. Relative size also small : Wing equal or shorter than the body, more rarely a little longer. Surface area of the wings is measured in one species of the genus Musidora and varies from 0.042 to 0.048 cm2. The load in these insects is equal to 0.019-0.032 gr/cm2. Relative weight of the wings is not known.
Shape of the wings
Very characteristic of the lifting not-costalized (musidoroid) type is the shape of the wings, having strongly convex anterior and posterior edges. The apex of the wing usually is pointed, more rarely blunt (many Streblidae). Basiala little expressed. Anal lobe often totally absent, more rarely (many Streblidae) it is present as the weakly convex hind-margin [of the wing]. Proportion of length and width of the wing equal to 1 : 2.25-3. The wings are long in the representatives of the Musidoridae, short in many Streblidae.
Skeleton of the wing
The most characteristic trait of this type is the absence of almost any trace of costalization : The veins are evenly distributed in the wing-blade, not shifted towards the anterior margin. Only the absence or weakening of the Costal vein at the wing's hind-edge is the very one sign [these wings do have] of costalization. The Subcostal vein is very short, flowing out into the anterior wing-edge very close to the base of the wing. Sometimes this vein is completely absent (Streblidae). Radial vein in the form of three simple branches of which the anterior often is shortened not reaching middle of fore-margin. Medial veins in the form of two (Streblidae) or three branches (Musidoridae). Cubital vein straight and long in Streblidae, and very short and curved in Musidoridae. Anal vein (1A) only present in Musidoridae. It is necessary to note that the described interpretation of the venation of these narrowly specialized diptera has, in a certain degree, a preliminary nature, and, probably, will be changed as a result of additional scrutinity. However, as to the objectives of the present enquiry, an exact homologization of the veins [i.e. a determination of their true identities] is not necessary.
The basiala lacks a fragma and has a free central membrane devoid of supporting elements. Cross-veins almost completely absent (Musidoridae), more rarely they are present, numbering two or three, on the wing-blade [that is the whole wing, apart from the basiala] (Streblidae). Chaetaria are inspected only in the genus Musidora and show themselves as convex not pigmented bristly hillocks.
Coverings of the wing
On inspected wings of Musidoridae there is a dense covering with fairly large microtrichia (11-15 [mu] ). Macrotrichia are distributed along almost all veins (without [such] bristles on only the second Radial branch and on some other veins) and along the wing's hind-edge. The length of the macrotrichia at the posterior edge varies from 45 mu near the wing-apex to 105 mu at the basal part of that edge. Still longer bristles are present at the hind-margin of the basiala.
The covering of the wings of the Streblidae, the latter not being known to me in nature, is, judging from drawings and descriptions, very similar to that of the Musidoridae. Scales are absent on the wings. Nervous system and sensory organs of the wing are not investigated.
Functional features
The life-history of the two groups of diptera possessing wings of the musidoroid type, is very different. The species of the family Musidoridae live among grassy vegetation in the moderate zone of the Earth. Their larvae feed on rotting leaves of various trees and live in the upper layer of the soil amidst vegetable remains. The flies run fast amidst the plants and are particularly common in shaded moist stations [habitats]. A completely different way of life we see in the species of the family Streblidae, being ectoparasites of bats. Almost the whole individual development of these insects takes place in the body of the mother-fly, and the latter gives birth to already mature larvae, that is, pupae (this family belongs to the artificial group of pupiparous diptera, Pupipara). In more detail the life-history of the Streblidae is not known to me. We must note the presence in this family of wingless forms, often very strongly changed as a result of the parasitic way of life.
Exact observations of the flight of the representatives of this type are absent. Judging from the structure of the wings and from the known living conditions, we may, amost with confidence, confirm the little significance of this function in the life-activities of these insects, and moreover, the very low mechanical qualities of their flight. This is completely evident in the case of parasitic streblids, and probably [merely probable] for musidorids. However, we cannot categorically ascribe a regressive nature to the flight-function of the musidorids. The absence of small-winged, and what is more, of wingless forms, the pecularity of the structure of the wings, and the free way of life, rather let us incline to an evaluation of the flight of the musidorids as a peculiarly narrow-specialized function, possibly carrying a regressive nature. A more precise evaluation of the flight-features in these flies would now be premature.
History and interrelationships of the type
We do not know of any fossil documents of the history of the type. The recent content of the type clearly points to the existence of two sharply separated subtypes. The first, the musidoroid-proper subtype, is characterized by wings pointed at the apex, the absence of cross-veins, the reduction [shortening] of the cubital vein, and the weakly developed Costal vein at the posterior wing-margin. Here do belong the few species of the one genus of the family Musidoridae.
The second subtype, the strebloid subtype, is characterized by wings rounded at their apex, the presence of the cubital vein and of two or three cross-veins, and the absence, along the posterior margin, of the Costal vein. To this subtype do belong species of the few genera of the family Streblidae, chiefly distributed in the tropics.
Determination of the genealogic relations of the musidoroid type with other types is far from easy. Peculiar is the reduced venation, almost devoid of traces of costalization, the simplified structure of the basiala, and, finally, the abundant development of macrotrichia -- all this points to an early origin of the type. The original forms of this type could not have possessed costalized wings. This fact does not allow us to derive the type from known subtypes of empidoid wings in which the original determining moment of evolution was the development of costalization. Apparently, source of the musidoroid subtype were certain forms of subtypes of empidoid wings not preserved anymore in the recent fauna, in which [empidoid subtypes] the chief direction of evolution of the wings consisted in their decrease of size as a result of the reduction of the flight-function (such as what we see in the corynetoid subtype !), whereby this process had begun very early on, when the venation was still rich in the ancestral forms of the Diptera. [See for a possible derivation of the wing-venation of Musidoridae from clythiod wings -- muscoid type -- Figure 3, above ]. This regressive development of the wings was determined by the absence of increase, or even decrease, of the wing-beat frequency, as a result of which [features of] costalization did not develop. The flight-function played a minor role in the locomotion of these insects. The regressive evolutionary direction of the wings, expressed by the musidoroid type, determined a certain similarity to exist between it and other, non-related, but also regressive forms of wings, namely with the primitive lifting (psychodoid) type, see Part LXIX . This similarity consists in the [more or less] similar shape of the wings [see HERE for a wing of the primitive lifting type, Psychoda sp., Psychodidae], uniformly broadened and even [in both cases] pointed at the apex, and in the similar distribution of parallel veins. Yet, a direct phylogenetic relationship between these types is completely excluded.
* * *
Lifting ultra-costalized (phoroid) functional wing-type
(Descriptions taken from ROHDENDORF, 1951, with additions and additional Figures)
Representatives of the type
Wings of the lifting ultra-costalized (phoroid) type are in the recent fauna possessed only by species of the family Phoridae (see next Figures). Here we should also mention the Jurassic family Palaeophoridae (bottom image of next Figure), being the direct ancestors of the first mentioned family.
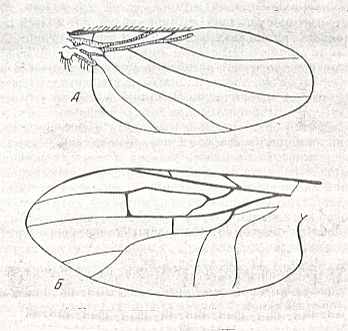
Figure 5 : Wings of representatives of the lifting ultra-costalized (phoroid) type.
Top image : Aphiochaeta sp., Phoridae. Length 3.5 mm. (After Rohdendorf, 1951).
Bottom image : Palaeophora ancestrix Rohd., Palaeophoridae. Jurassic of Karatau (Southern Kazachstan). Length a little less than 2 mm. (After Rohdendorf, 1938) [ In 1938, in Trudy Paleontologicheskovo Instituta, Tom VII, Issue 3, Rohdendorf had described this fossil -- nr 2452/329 -- as Archiphora ancestrix . The name had to be changed because of the priority of the name Archiphora (see next Figure) ].
(From ROHDENDORF, 1951)
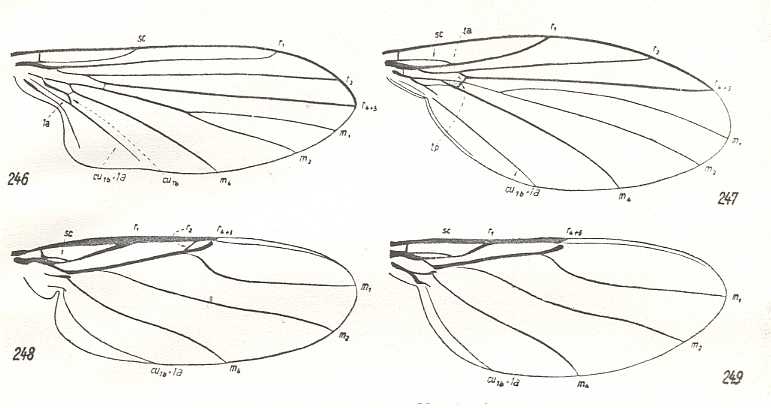
Figure 6 : Venation of Phoridea, compared with that of Clythiidae.
246 - Opetia nigra Meig., Clythiidae. (After Schmitz, 1929). Clythiod subtype of muscoid type.
247 - Sciadocera (Archiphora) patagonica Schmitz, Sciadoceridae. (After Schmitz, 1929). Sciadoceratoid subtype of empidoid type.
248 - Aneurina thoracica Meig., Phoridae. (After Hennig, 1954). Phoroid type.
249 - Phora stictica Meig., Phoridae. (After Hennig, 1954). Phoroid type.
246 ==> 247 ==> 248 ==> 249 forms a neat derivational line.
(From HENNIG, 1954)

Figure 7 : Phora aterrima Fabr. Phoridae. about 4 mm.
(After CHINERY, in Elseviers insektengids voor West-Europa, 1983)
Size of the wings
Absolute size of the wings small, varying between 1.5 and 5 mm. Relative size about equal to body-length. Surface-area of the wings is known only of a few representatives of the family Phoridae and varies between 0.023 cm2 and 0.060 cm2. Weight of wings not determined. Load in the investigated species varies between 0.022 gr/cm2 and 0.037 gr/cm2.
Shape of the wing
Anterior wing-edge almost straight, weakly convex. Wing-apex not clearly individualized [i.e. not clearly standing out], very large [as such causing its not-standing-out]. Posterior wing-edge rather strongly and uniformly convex. Anal lobe very large, almost rectangular, sharply separating off the broad basiala. Wings broad, only 2-2.5 times longer than wide.
Skeleton of the wing (see Figure 5, top image)
Most characteristic of this type is the extreme degree of costalization, which consists in the shift and thickening of the veins of the anterior margin, in the lightening [i.e. freeing of veins or parts of them, or weakening of them] of a large part of the posterior marginal, middle, and apical areas of the wing, by way of reduction of veins, and, finally, in the enlargement of the anal lobe. Costal vein far from reaching the wing-apex, often running only up to the middle of the anterior margin. Subcostal vein shortened, in the form of a small outgrowth, freely ending up in the membrane of the costal cell, close to the humeral cross-vein [or Subcosta ending up in the first Radial vein, see 248 and 249 of Figure 6 ]. First Radial vein strong, the second of a complex nature, thicker than the first, at its end with a fork, and ending up at the apical end of the Costal vein. The remaining four veins in the form of weak, irregularly curved, simple branches, running from the wing-base and the strong trunk of the posterior Radial vein to the wing's hind margin. In fact strong venation is limited to the basal half of the anterior part of the wing. The remaining wing-blade almost not strengthened.
Basiala well standing out only posteriorly by the border of the anal lobe. Anteriorly the borders of the basiala are not clear. The most massive supporting part of the basiala is its chief elevated element, the base of the Radial vein, which has acquired a tube-like form, not forming a phragma. The costal margin of the basiala is also fairly thick. The posterior margin of the basiala is formed by a thickened vein (the Jugal vein?) containing a trachea -- the direct continuation of the marginal trachea of the wing-scale. The second elevated element of the basiala (the handle of the second Anal vein) is small and in essence is just a support of the posterior chaetarium. The anterior (radial) and posterior (anal) chaetaria are present, but not clearly standing out. The lower element of the basiala -- the handle of the Cubital and Anal veins -- is present, but not sharply distinct from the posterior elevated element.
Coverings of the wing
The whole wing is densely covered with microtrichia, the length of wnich is small (5-10 mu). On a large part of the surface of the wing macrotrichia are absent. Only along the anterior wing-edge, that is, along the Costal vein, and along the posterior margin of the basiala (at the place of the absent alula) there are long and strong bristles, placed in one or sometimes two (along the Costal vein) series. Sometimes there is a single bristle at the basal part of the posterior Radial branch. The structure of these big wing-macrotrichia is very characteristic : They are covered with thin hairs, they are f e a t h e r - l i k e. Scales are absent on the wing [blade]. Nervous system and sense-organs on or in the wing are not studied.
Functional features
The way of life of the representatives of the family Phoridae are little known. Many species have larvae living in animal excrements, in decaying plant- and animal-matter, in carrion. Many forms are known that are co-dwellers with ants and with other social hymenoptera, or are predators or parasites of certain other insects. Winged phorids are encountered in the most diverse stations [habitats] -- amidst grassy and shrubby vegetation, on flowering plants, on the soil, on rotting wood, on windows of rooms, and so forth. Exact observations as to the way of life, and especially the flight of these insects are, up to now [1951] absent. These flies swiftly run on the substrate, reminding us a little, as to their behavior, of certain fungivoroids. They eagerly take flight, although the chief way of locomotion is, apparently, running. I had no opportunity to carefully observe the flight of phorids, and it remains to judge about it only on the basis of the morphological features of these flies. The minute size (weight seldom being above 0.002 gr), the extreme costalization of the wings, the powerful thorax -- all this bears witness to a high wing-beat frequency making a well-governable flight possible with a large "store of power", that is, to [these flies] perfectly moving in the air in limited spaces [in narrow places, that is], for example slowly "hovering", while making diverse turns at low speed. The significance of such a kind of flight in the life-activities of the insect is still completely unclear : May be, the absolute weight of these flies during their individual life strongly varies as a result of the maturing of sex-products of the females, to which some indirect data point. On the whole, however, this whole question, concerning the meaning of the characteristic flight of the phorids and [concerning] the structure of their wings, remains open. The powerful development of their legs, the presence in this group of wing-less forms -- all this calls for a cautious assessment of the evolutionary significance of the phoroid type. Apparently, in this case we have to do with an example of narrow specialization of the flight-function, the evolution of which went into the direction of a one-sided improvement of the quality of flight, extreme increase of wing-beat frequency [judging from the extreme degree of costalization] having increased lifting-power and speed of take-off as a result of the decrease of absolute speed of flight.
History and transformations of the type.
The origin of the type is still unclear. Existing fossil material (a -- in Jurassic deposits of Karatay -- discovered representative of the family Palaeophoridae)
In the year 1938 a new genus Archiphora (type of genus : Archiphora ancestrix Rohdendorf, 1938) was described by me from the Jurassic of Karatau. In the year 1929 Schmitz had described a recent south-american genus Archiphora (type : A. patagonica Schmitz, 1929). Therefore, the name given by me to the Jurassic genus must be replaced by another. I propose it to rename Palaeophora nom. nov. Automatically is changed also the name of the family, Archiphoridae, into Palaeophoridae.
throws some light on the way of the formation of the type. The wings of the family Palaeophoridae possess a more rich venation than those of the recent Phoridae, although the basic traits of the phoroid type (shift and thickening of the Radial veins) were already expressed. Naturally, this Jurassic form may be distinguished as belonging to the special palaeophoroid subtype (maybe still belonging to the empidoid type?). Recent and Tertiary phorids make up the phoroid-proper subtype.
Source of the phoroid type undoubtedly were forms with wings of the empidoid type whereby, apparently, costalization in them was already sufficiently expressed, as in, for instance, the microphoroid subtype.
Phoroid wings [i.e. wings of the phoroid type] illustrate a progressed stage of improvement of the wings into the direction of increase of wing-beat frequency, increase of lifting-power, with small absolute size of the insect. The insufficient knowledge of the features of the life-activity of the phorids does not allow to clarify directions and causes of the realization of the recent traits of the lifting ultra-costalized (phoroid) type of wings. Only in an overall, complete, analysis of all aspects of the organization of these insects will it be possible to clarify this question. But this, however, already goes beyond the framework of the present investigation.
* * *
Traction-lifting many-veined (syrphoid) functional wing-type
( Descriptions taken from ROHDENDORF, 1951, with additions and additional Figures) [In the Russian text the type is named : traction-lifting lightly-veined. But looking at most representatives of this type we see that all the wings are, on the contrary, many-veined. So we will call this type : traction-lifting many-veined.]
Representatives of the type
The large family Syrphidae (hover-flies proper) and the small group Dorylaidae possess wings structured according to the traction-lifting many-veined (syrphoid) type. [ A good reason to include the Dorylaidae (= Pipunculidae) into the present type is their well-developed ability to hover, just like the Syrphidae.] See Figures 8, 9, 10, and 11, below.
A - Chalarus holosericeus Meig., Dorylaidae.
Length 3.2 mm.
dorylaioid subtype
|
B - Dorylas silvaticus Meig., Dorylaidae.
Length 3.2 mm.
dorylaioid subtype
|
C - Syrphus sp., Syrphidae.
Length 11 mm.
syrphoid-proper subtype
|
D - Volucella pellucens L., Syrphidae.
Length 14 mm.
syrphoid-proper subtype
|
|
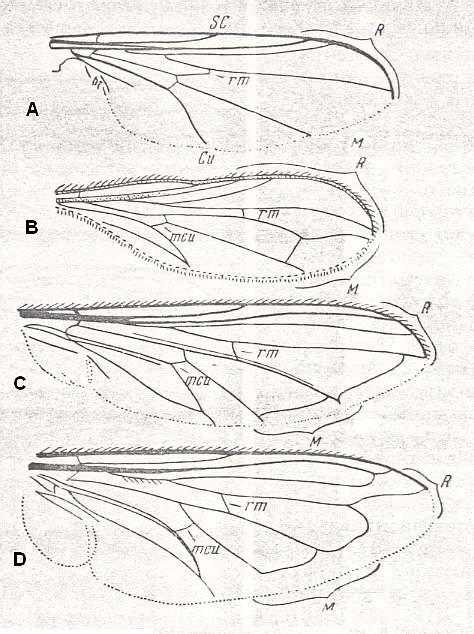
(After ROHDENDORF, 1951)
|
Figure 8 : Wings of representatives of the traction-lifting many-veined (syrphoid) type.
E - Eumerus sp., Syrphidae.
Length 5.5 mm.
syrphoid-proper subtype
|
F - Spilomyia diopthalma L., Syrphidae.
Length 13.5 mm.
syrphoid-proper subtype
|
G - Microdon eggeri Mik., Syrphidae.
syrphoid-proper subtype
|
|
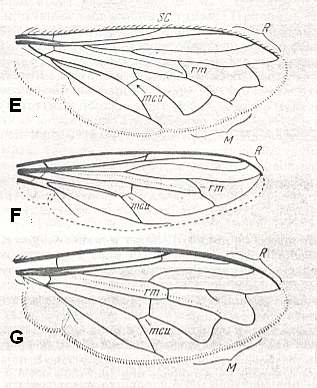
(After ROHDENDORF, 1951)
|
Figure 9 : Wings of representatoves of the traction-lifting many-veined (syrphoid) type.
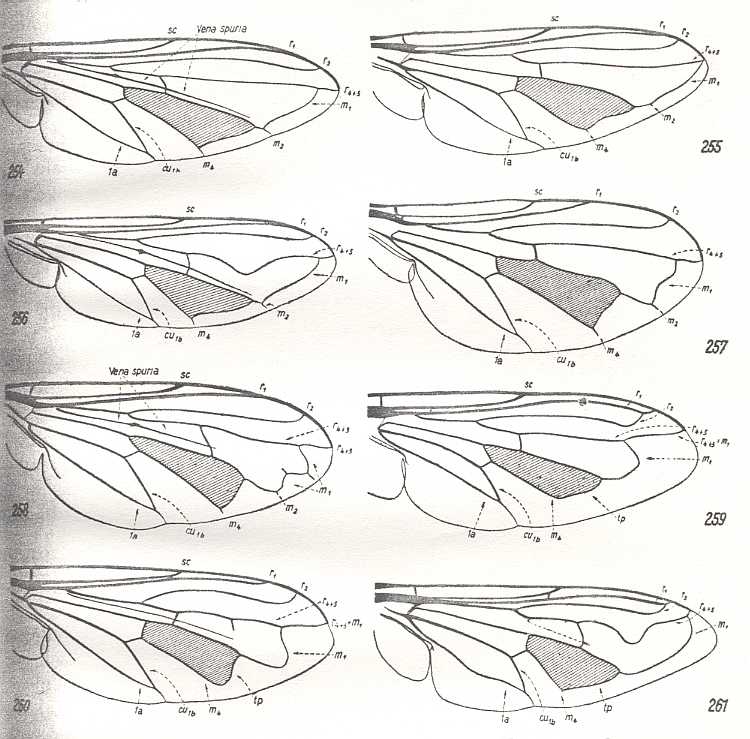
Figure 10 : Wing-venation of representatives of the traction-lifting many-veined (syrphoid) type (syrphoid-proper subtype).
| 254 - Syrphus ribesii L., family Syrphidae.
|
255 - Nausigaster haemorrhoidalis Phil., family Syrphidae.
|
256 - Didea fasciata L., family Syrphidae.
|
257 - Pipizella virens Fabr., family Syrphidae.
|
| 258 - Eumerus latitarsis Macq., family Syrphidae.
|
259 - Volucella bombylans L., family Syrphidae.
|
| 260 - Microdon devius L., family Syrphidae.
|
261 - Lathyrophthalmus aeneus Wied., family Syrphidae.
|
(After HENNIG, 1954)

Figure 11 : Representatives of the traction-lifting many-veined (syrphoid) wing-type (syrphoid-proper subtype).
| 10 - Volucella inanis L., about 20 mm. Family Syrphidae.
|
11 - Scaeva pyrastri L., about 15 mm. Family Syrphidae.
|
12 - Syrphus ribesii L., about 12 mm family Syrphidae.
|
13 - Milesia crabroniformis Fabr., about 25 mm. Family Syrphidae.
|
| 14 - Leucozona lucorum L., about 12 mm. Family Syrphidae.
|
15 - Doros conopseus Fabr., about 16 mm. Family Syrphidae.
|
(After CHINERY, in Elseviers insektengids voor West-Europa, 1983)
Size of the wings
Absolute size of the wings is medium, often large, seldom minute, from 2 mm (some Dorylaidae) up to 15-20 mm. Relative size of the wings small, as a rule less than length of body, more rarely equal to it, very seldom a little longer than the length of the body (many Dorylaidae). Surface-area of the wings known only of a series of species of Syrphidae and varies between 0.123 cm2 (minute species Melanostoma mellinum L.) and 1.219 cm2 (the large Volucella pellucens L.). Relative weight of the wings of syrphids not large and varies between 0.41 and 1.20 percent of the weight of the whole insect. Load is also measured only in some syrphids and varies between 0.014 and 0.261 gr/cm2.
Shape of the wings
Anterior wing-edge up to the wing's apical fourth straight. The apex is neatly expressed. Hind-edge of wing evenly and rather strongly convex. Anal lobe large, seldom only little projecting (Dorylaidae, Figure 8 - A, B). Alula very large and long in the representatives of the Syrphidae ( Figure 8 - C, D and Figure 9 - E, F, G ), short and little projecting in Dorylaidae. Wings elongate, always not less than 2.5-3 times longer than wide.
Skeleton of wing
Most characteristic of this type are : the presence in the basiala of a well developed phragma, very rarely weakened and represented by a firm fold, the development of an insignificantly costalized venation covering almost all of the wing, reduction of the number of Medial veins, and, finally, the large size of the cubital cell. The Costal vein always ends at the wing-apex, and is reduced at the posterior wing-margin. Subcostal vein usually long, often ending up in the Costa beyond mid-wing. Three Radial veins, which are simple branches, always ending up at the anterior wing-margin or at the wing's apex, sometimes partly coalescing with each other. Medial veins in the form of two branches, enclosing a characteristic intermedial cell, distally bordered by the intermedial cross-vein often acquiring the form of a continuation of the posterior Medial branch. Seldom is the anterior Medial branch reduced (genus Chalarus from Dorylaidae, Figure 8 - A ). The anterior Medial vein is of complex origin, composed of two anterior branches, coalesced into one common vein. Sometimes there is at the place of its bend an outgrowth, pointing to the double nature of this vein. Cubital and Anal veins long, unifying at their end, forming the characteristic sharp cubital cell. Seldom is the Anal vein in its distal end reduced (genus Chalarus). The cross-vein r-m almost always placed "normally", more rarely shifted towards the distal part of the wing, lying obliquely (some Syrphidae, Figure 9 F ). The posterior edge of the wing has a zone free of veins in the form of a fringe, separated from the anal lobe by the end of the first Anal vein, which runs to the wing-margin. Sometimes this marginal fringe is subdivided by the ends of the Medial veins, also running to the wing's margin. Seldom, in addition to the cross-vein r-m, there are rudiments of other radio-medial cross-veins (genus Microdon, Figure 9 G ). The cross-vein m-cu is placed obliquely, turning -- as often happens in diptera -- into the basal section of the posterior Medial branch. The cross-vein r-m cuts through a longitudinal fold, the so-called "pseudo vein" (vena spuria) -- a characteristic feature of all representatives of the family Syrphidae. A similar fold is sometimes also present in the first radial cell. The presence of the "pseudo vein" was widely used in all kinds of enquiries concerning the homology of veins [i.e. their true identity with respect to the primitive prototypic venation of diptera] of Syrphidae and other related families. Attributing this element of the venation the nature of a rudiment of a disappeared vein of a more rich venation of ancestral forms, i.e. the interpretation of the vena spuria as some sort of left-over, has radically altered the terminology of the venation of these diptera. Not clarifying more precisely this issue, I note only the important functional significance of the vena spuria as a strengthened fold, placed in the central area of the wing. Such an extra support of this part of the wing-blade is undoubtedly connected with the overall configuration of the whole venation of these little-costalized wings. There is no serious basis to ascribe to the "pseudo vein" the nature of a rudiment, a left-over of an ancient wing-venation. It is far more natural to see this pseudo vein as a new-formation, a strengthening fold [the Russian text reads : a fold which is strengthened], the appearance of which is determined by the whole mechanics of the wings of the syrphoid type.
The basiala clearly stands off, and is provided with two elevated elements ( [bases of the] Radial and second Anal vein) and a lower central element -- handle of Cubital and first Anal vein. This lower element is always large, well expressed. Phragma, as a rule, firm and often coalesced with the strengthened and colored central field of the basiala. The handle of the second Anal vein is large and thickened, and carries a chaetarium (as also does the radial handle). Wing- and thoracic-scales, as a rule well developed, more rarely short and rudimentary ( Dorylaidae, Figure 8 - A [the mentioned feature -- short and rudimentary scales -- is not particular visible in the Figure referred to] ).
Coverings of the wing
Wings covered, as a rule, completely with densely packed minute microtrichia. Seldom microtrichia are absent in certain places of the membrane, usually in the middle and proximal parts of the wing and on the alula (for instance in representatives of the genus Syritta). Macrotrichia are absent on the membrane, and developed only in the Costal, and seldom on the first Radial vein. The posterior margin of the wing, and especially the anal lobe and alula, are provided with often fairly long macrotrichia. Wing- and thoracic-scale carry long thin hairs at the margin. No scaly covering [like in butterflies] has been developed on the wings. Nervous system and sense organs [on and in the wings] have not been investigated by me.
Functional features
The life-history of syrphids and dorylaids is known in general terms.
The larvae of syrphids develop under the most diverse conditions -- in water-basins, decaying vegetable materials, in tissues of living plants, in dung, consuming living aphids, and, finally, living in ants' nests. The larvae of dorylaids are specialized internal parasites of various cicadas. The winged insects of both families spend the greater part of their life in flight, visiting flowering plants, on which they chiefly feed on pollen. Flight of syrphids has been especially investigated (the reason for this is the abundancy and wide distribution of these insects in all habitats, including those of humans) -- these flies were used to anwer the question of wing-beat frequency. Thus, the wing-beat frequency in species of the genus Syrphus varies from 131 to 170, in Eristalis from 177 to 193, and in Volucella was equal to 179 (according to Voss, in Rohdendorf, 1949, p. 47). In addition, with a species of the genus Volucella the trajectory followed by the wing during wing-beat was determined. The high degree of perfection of the flight of syrphids is well illustrated by the ability of these insects to "hover" while staying at the same point. Such a kind of flight is known of representatives of a series of genera of the family -- Syrphus, Eristalis, and others. The speed of flight is undoubtedly very high, although up to now not precisely investigated. Simple visual observations point to a high speed of flight of many large syrphids (genus Eristalis, Syrphus, Milesia, and others), which, when disturbed, quickly fly off far away from the flowers. In addition, these flies are able to slowly and freely fly around the flower in a comparatively small amount of space. The significance of flight in the life-activities of syrphids is unconditionally very great : all these flies need to feed on flowers, possess weak legs, not making possible fast running, and completely unprotected, lacking a stinging snout. The high quality of flight has important significance in their life.
Flight in Dorylaidae is almost completely unstudied. Existing observations point to a slow, but prolonged flight above shrubs, wherby the insects for only a short time settle on the vegetation. Visiting flowering plants is not the rule -- probably feeding of the winged phase is not obligatory, by which these flies are distinguished from syrphids. The structure of the wings, their greater [relative] size, the weak development of the anal lobe and of the appendages of the basiala, -- all this bears witness to lower qualities of the flight-function of the dorylaids as compared with syrphids, or, more precisely, to a different nature of their flight.
Evaluating the features of the wings of the representatives of the syrphoid type, we must note the high supporting qualities of the basiala, on the one hand, and the low degree of costalization of the wing-blade, on the other. These traits bring the syrphoid wings close to those of the tabanoid type. The reasons for this undoubtedly boil down to the same : the large absolute size of these insects. Like in the tabanoid type a rich venation having an important supporting function in large wings, so also in the syrphoid type the wings are little-costalized for the same reason -- the necessity to preserve firmness with increasing wing-size. Mechanical similarity of the syrphoid wings with other wings is best of all seen in the midaoid subtype [10th subtype of the tabanoid type] and partly in the nemestrinoid subtype (11th subtype of the tabanoid type). This similarity consists in the development of a characteristic marginal fringe of the posterior margin of the wing. The presence of a vein-free fringe along the posterior wing-margin indicates a certain degree of costalization of the venation, more precisely, an increase of wing-beat frequency, providing the insect to obtain a high degree of governability in flight, in low speeds. The overall large size of the wings, their elongate nature, and firmness, guarantee, in addition to a high degree of governability, also a high absolute speed of flight.
History and transformations of the type
Up until now fossil remains of representatives of the syrphoid type of wings are only known from Tertiary deposits. Such are the many species of a whole series of genera, in their majority also present in the recent fauna. Studying these Tertiary syrphids does not allow to clarify the interrelationships of this type with other types. Only a comparative-anatomical investigation of recent forms can determine the ways of evolution of the syrphoid wings. The large majority of representatives of the type belongs to the family Syrphidae, and may be separated in the form of the special syrphoid-proper subtype, of which large size (as a rule not less than 6 mm), a well developed anal lobe, alula, and wing- and thoracic scale, are characteristic. In addition, the wings of this subtype are characterized by small [relative] size, not surpassing the length of the body, -- usually they are much smaller. Not less characteristic of this subtype are the well developed Medial veins -- development of a vein-free fringe at the posterior margin, and rich venation. The representatives of this subtype are fairly diverse, whereby some characteristic directions of specialization of the wings are found (their elongation in the group Milesiinae, Figure 9 - F and an increase of their size and their broadening in Volucellinae, Syrphinae, Figure 8 - C, D , accompanied by alterations of the venation. However, all these [just mentioned] forms of wings of syrphids have a subordinated significance and do not deserve to comprise yet another subtype.
The second well distinguished subtype of syrphoid wings, characteristic of representatives of the family Dorylaidae, may be called the dorylaoid subtype [see Figure 8 - A, B ]. Of this subtype small absolute (up to 2mm), and, on the contrary, large relative size of the wings, always longer than the length of the body, is characteristic. The wings are narrowed at the base and possess a weakly expressed anal lobe, a rudimentary alula and [wing-]scale. A marginal [vein-free] fringe is absent, the whole venation of the wing being "weak" (veins thinner). Sometimes processes of reduction of the venation take place, consisting in the disappearance of the anterior Medial branch and the distal section of the Anal vein (genus Chalarus, Figure 8 - A ).
The origin of the traction-lifting many-veined (syrphoid) type is more or less clear : Its source were the most simple forms of muscoid wings, belonging to the clythioid subtype. The direction of evolution having led to the recent forms of the syrphoid type apparently consisted in processes of increase of absolute body-size and improvement of flight (its speed). Primitive forms of the clythioid subtype possessed only little-expressed features of costalization (decrease of the number of Medial and Radial branches, and the [nature of the] posterior marginal area of the wing [shortening of the cubital cell] ) and a little-perfected basiala. On the other hand, comparing the features of the syrphoid and tabanoid wings, we may definitely see the approximate similar evolutionary significance of these types, which are among the most progressive perfect forms of flight-apparatus in diptera.
* * *
This concludes not only the exposition of the traction-lifting many-veined (syrphoid) functional wing-type, but also our exposition (having followed Rohdendorf, 1951) of all the functional types and subtypes in the Order Diptera. And, in addition, we now have also concluded our investigation of the two insect Orders Hymenoptera and Diptera. This investigation was about their evolution as driven by ecological factors and in the context of the metaphysics of the Implicate and Explicate Orders. This metaphysics is expressed by our noëtic theory of evolution (i.e. organic evolution expounded in terms of the interaction between the Implicate and Explicate Orders), and this theory was gradually developed on this Part of Website (V) while investigating the evolution of Diptera and Hymenoptera. There we had learned the polyphyletic development, not only of the mentioned insects but of all insects, and indeed of all living beings whatsoever. Organic species are materialized "strategies-to-exist-in-the-Explicate Order", and these strategies are forged in the Implicate Order driven by the 'desire' of immaterial patterns to become material, i.e. to atain ontological completion. Projection of such a strategy into the Explicate Order depends on existing existential conditions there, that is, the existence of vacant ecological niches.
Well, in order to test and develop our noëtic theory of evolution still further, we must expand our evolutionary investigations to at least all insects. That will bring in paleo-entomology, fossil insects. And because the latter are mainly found in the form of wing-impressions only (whole insect-bodies are rarely well-preserved), and because the study of wings and flight-function in insects has already began to pay off in this and all the previous documents on insect evolution, we will, when studying all insects from an evolutionary viewpoint, keep on concentrate on wings and flight-function. But we will do this in the next Part of Website, that is, in Sixth Part of Website . Also in that Sixth Part we will further work out our noëtic theory of evolution : Especially in the "Theoretic Intermezzo" there (after part VIII ) we give an extensive summary and further elaboration of this theory.
e-mail :  ( Please write in ' Subject ' entry : ' METAPHYSICS ', in order for me to be able to distinguish your mail from spam )
( Please write in ' Subject ' entry : ' METAPHYSICS ', in order for me to be able to distinguish your mail from spam )
To continue, click HERE for Sixth Part of Website : Insect Evolution (paleo-entomology)..
Back to Homepage
Back to Contents
Back to Evolutionary Part XIV
Back to Evolutionary Part XV
Back to Evolutionary Part XVI
Back to Evolutionary Part XVII
Back to Evolutionary Part XVIII
Back to Evolutionary Part XIX
Back to Evolutionary Part XX
Back to Evolutionary Part XXI
Back to Evolutionary Part XXII
Back to Evolutionary Part XXIII
Back to Evolutionary Part XXIV
Back to Evolutionary Part XXV-A
Back to Evolutionary Part XXV-B
Back to Evolutionary Part XXV-C
Back to Evolutionary Part XXVI
Back to Evolutionary Part XXVII
Back to Evolutionary Part XXVIII
Back to Evolutionary Part XXVIII-A
Back to Evolutionary Part XXIX
Back to Evolutionary Part XXX
Back to Evolutionary Part XXXI
Back to Evolutionary Part XXXII
Back to Evolutionary Part XXXIII
Back to Evolutionary Part XXXIV
Back to Evolutionary Part XXXV
Back to Evolutionary Part XXXVI
Back to Evolutionary Part XXXVII
Back to Evolutionary Part XXXVIII
Back to Evolutionary Part XXXIX
Back to Evolutionary Part XL
Back to Evolutionary Part XLI
Back to Evolutionary Part XLII
Back to Evolutionary Part XLIII
Back to Evolutionary Part XLIV
Back to Evolutionary Part XLV
Back to Evolutionary Part XLVI
Back to Evolutionary Part XLVII
Back to Evolutionary Part XLVIII
Back to Evolutionary Part XLIX
Back to Evolutionary Part L
Back to Evolutionary Part LI
Back to Evolutionary Part LII
Back to Evolutionary Part LIII
Back to Evolutionary Part LIV
Back to Evolutionary Part LV
Back to Evolutionary Part LVI
Back to Evolutionary Part LVII
Back to Evolutionary Part LVIII
Back to Evolutionary Part LIX
Back to Evolutionary Part LXa
Back to Evolutionary Part LXb
Back to Evolutionary Part LXc
Back to Evolutionary Part LXI
Back to Evolutionary Part LXII
Back to Evolutionary Part LXIII
Back to Evolutionary Part LXIV
Back to Evolutionary Part LXV
Back to Evolutionary Part LXVI
Back to Evolutionary Part LXVII
Back to Evolutionary Part LXVIII
Back to Evolutionary Part LXIX
Back to Evolutionary Part LXX
Back to Evolutionary Part LXXa
Back to Evolutionary Part LXXb
Back to Evolutionary Part LXXI
Back to Evolutionary Part LXXI
Back to Evolutionary Part LXXII
Back to Evolutionary Part LXXIIa
Back to Evolutionary Part LXXIII
Back to Evolutionary Part LXXIIIa
Back to Evolutionary Part LXXIV














 ( Please write in ' Subject ' entry : ' METAPHYSICS ', in order for me to be able to distinguish your mail from spam )
( Please write in ' Subject ' entry : ' METAPHYSICS ', in order for me to be able to distinguish your mail from spam )