

e-mail :
 ( Please write in ' Subject ' entry : ' METAPHYSICS ', in order for me to be able to distinguish your mail from spam )
( Please write in ' Subject ' entry : ' METAPHYSICS ', in order for me to be able to distinguish your mail from spam ) Following ROHDENDORF, 1964, pp. 34, we discuss the infraorder Deuterophlebiomorpha. We do this wholly in terms of the family Deuterophlebiidae which is the only family known in this infraorder.
Extent, evolution, and system
Of this family five species of a single genus Deuterophlebia EDWARDS are known. They are distributed in several regions of the mountain systems of the Northern hemisphere -- Altai, Tian-Shan, the Himalayas, Japan, and the Rocky Mountains of North America.
Having been for the first time described in 1922, these insects up to now remain insufficiently studied. Most of the data are about the organization of the larva. Fossil remains are still unknown.
The deuterophlebiid diptera are characterized by their very isolated position in the taxonomic system of the Order, while having some features similar to those of Blephariceridae (way of life and organization of the larvae) and partly to those of Chironomidea (some features of the antennae of all phases and the spiracles of the pupae). The idiosyncrasy of the Deuterophlebiidae forces us to suppose their very early origin from the common forms of Tipulomorpha and Blephariceromorpha.
Chief features
The characteristic way of life in conditions of high-mountain terrain and the larvae living on rocks of swiftly-flowing mountain streams, determines the features of these evidently relict forms. These features are the same in the Blephariceridae which together with them live in precisely the same conditions of fast-flowing mountain streams.
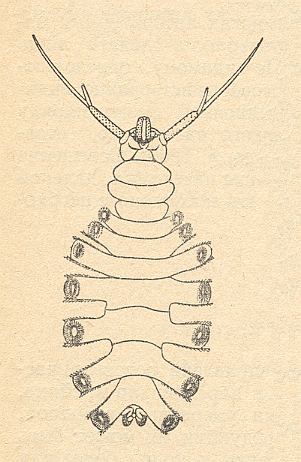
Figure 1 : Larva of Deuterophlebia japonica KITAKAMI, ventral view.
(After KITAKAMI, from HENNIG, 1948-1952, from ROHDENDORF, 1964)
The larvae (see Figure above) have a weakly expressed concentration of the body-segments. They have a free head with enormous antennae, a 3-segmented thorax lacking appendages, and an 8-segmented abdomen, which carries on lateral protuberances of the first seven segments strong circular suckers, making it possible for the larva to live and move about in conditions of fast flowing water. The mouthparts of the larva (complex structure of the upper jaws) let us suppose that they feed on different overgrowths on rocks in streams (algae and unicellular organisms?), which the larva may collect and filter. The respiratory system of the larva is closed, and gas-exchange apparently takes place by means of special cutaneous protuberances, [that is,] gills on the last abdominal segment.
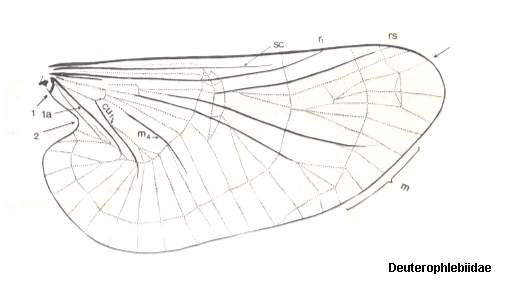
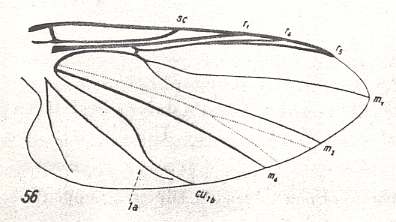
Figure 2 : Wing of Deuterophlebia spec. from Sikkim (Further India). ( Deuterophlebiidae).
The arrow at the wing-tip indicates the location where the Costa turns pale.
(After HENNIG, 1968)
The wings are very large (see Figure above), and together with the strong convexity of the thorax bears witness to the development of dorsoventral muscles -- lifting the wings -- and, consequently, to a high wing-beat frequency and the development mainly of traction-force.
Conflicts and determining tendencies in the historical development.
The basic conflict in the history of the Deuterophlebiids determing their recent state is expressed by desimaginization and aphagia of the winged insects. The refined feeding of the larvae in water-basins, the severe living-conditions of the winged phase (altitude, deficiency of food), and changes of temperature, led to the working-out of a non-feeding short-lived adult insect. The determining tendency in the solution of this conflict was the refinement of feeding of the larva in conditions of a fast mountain stream : The organization of the larva indicates that this protected insect is able to firmly hold itself and move about by means of its abdominal pseudopods in conditions of very fast moving water while remaining to be able to actively collect food from the substrate as a result of the free anterior end of the body (thorax and head). In trying to uncover the more remote history of the Deuterophlebiids, to evaluate the main features of their organization and understand the essence of the conflicts that have determined their historical development, one must first of all note, on the one hand, the free way of living of the larva [instead of entirely living in the depths of its nutritive substrate], which does not have a worm-like shape, and eats while crawling over the surface of the substrate, and, on the other hand, the great refinement of the flight-apparatus, antennae, and prehensile legs of the winged insect. All these features bear witness to the chief conflict in the history of the Deuterophlebiids, consisting in insufficient food for the larvae on the surface of the substrate and their great vulnerability to enemies (first of all, predators). The working-out of becoming a stenobiont organism, the withdrawal of the larvae into those parts of the stream with the swiftest current, made up the determining tendency to resolve this conflict. The winged insects developed high powers of flight for colonization [ = locally spreading the species] and the realization of reproductive activity, while feeding and life-span of the adult were shortened.
This short exposition of the features of this rare and relict group is of course just provisional and only sketches the most general features of its history.
Conclusions
The clearly expressed relict diptera Deuterophlebiomorpha had worked out desimaginization and a refinement of the larval phase having become a well-protected stenobiont, living in extreme conditions of fast-flowing streams in high mountains. The short-lived winged insect is characterized by aphagia, retrograde development of the digestive organs together with a highly-developed flight-apparatus and sense organs.
The insufficiency of data concerning the feeding of the larvae, the way of life of the winged insects, and reproduction, do not make it possible to more exactly describe the pathways of development of this rare and peculiar group of insects.
Having reproduced ROHDENDORF's exposition about the possible historical development of the Deuterophlebiidae, we can add, from other sources, some more data :
RICHARDS & DAVIES, 1977, Imms' General Textbook of Entomology, place the family Deuterophlebiidae, together with the families Dixidae, Culicidae (mosquitoes), Simuliidae, Thaumaleidae, Ceratopogonidae, Chironomidae, and Blephariceridae (to be dealt with in the next document), into the superfamily "Culicoidea", and characterize the family Deuterophlebiidae as follows :
Antennae filiform, very elongate. Wings with a network of creases. Ocelli [non-compound single eyes, in many insects present in addition to two large compound eyes], mouthparts, and true venation absent.
This small family consists of a single genus, Deuterophlebia, which is perhaps allied to the Blephariceridae and occurs in N. America, Japan, and the mountains of C. Asia. Pennak (1945) has revised the group and summarized its biology. The larva has seven pairs of large segmental outgrowths bearing suckers and posteriorly what have been described as anal blood-gills.
HENNIG compared the wings of the Deuterophlebiidae with those of the Blephariceridae (representing ROHDENDORF's infraorder Blephariceromorpha, dealt with in the next document) and apparently succeeded in identifying the few true veins in their wings. He did this in : Stuttgarter Beiträge zur Naturkunde, Nr. 193, 1968, Kritische Bemerkungen über den Bau der Flügelwurzel bei den Dipteren und die Frage nach der Monophylie der Nematocera.
Figure 3 :
Top image : Wing of Liponeura bilobata LOEW. ( Blephariceridae).[Click twice on image to have it magnified and improved]
Bottom image : Wing of Deuterophlebia spec. from Sikkim (Further India). ( Deuterophlebiidae). The arrow at the wing-tip indicates the location at which the Costa turns pale. [Click twice on image to have it magnified and improved]
(After HENNIG, 1968)
According to me [HENNIG] it is not all too difficult to compare the wing-venation of the Deuterophlebiidae (see Figure 3, bottom image) with that of the Blephariceridae (see Figure 3, top image). The following points appear important to me :
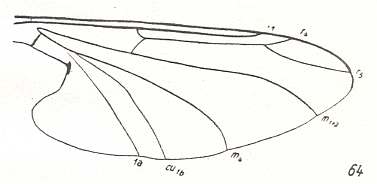
Figure 4 : Wing-venation of Hapalothrix lugubris LW. ( Blephariceridae).
(After HENNIG, 1954)
From such a wing one can easily formally derive that of Deuterophlebiidae : Apart from a more strongly enlargement of the anal lobe and a broadening of the middle part, we see especially an "expansion" of the wing region lying between the Radial Sector and the anterior branch of the Media on the one hand, and between the posterior branch of the Media-proper and the anterior branch of the Cubitus (=M4) on the other (see Figure 3, bottom image ). [We must realize that the Cubitus consists of two master branches, viz., the anterior master branch CuA, or, if one prefers to write, Cu1, and the posterior master branch CuP, or, if one prefers to write, Cu2, which in Diptera is either just a groove in the membrane or (secondarily) strongly reduced or absent. Now it is supposed that the anterior master branch of the Cubitus (CuA, Cu1) is itself forked, forming the characteristic "cubital fork" which consists of Cu1a (anterior branch) and Cu1b (posterior branch). And while the original M5 (fifth master branch of the Media) of the ancestral mecopterous wing has (in dipterous wings) long been disappeared or shifted to the extreme wing-base, M4 (fourth master branch of the Media) is still, in a way, present : Its base (in diptera representing the upper part of the cross-vein tb (or m-cu)) reaches the anterior branch (Cu1a) of the "cubital fork" and then coalesces entirely with it, and in this way together -- as one single vein -- reaching the wing-margin. This interpretation of the cubital region of the dipterous wing is HENNIG's and is expressed in his Figure (figure 5, in his 1954 article)] :

Figure 5 : Scheme of ground-plan of dipterous wing-venation, based on, and inspired by, the wing-venation of Protoplasa fitchii O.S. ( Tanyderidae).
(After HENNIG, 1954)
4. In Blephariceridae (see Figure 3, top image ) the Radius has shifted closely to the Costa and both veins are in places thickened. The Subcosta, lying between them, is significantly shortened. In the depicted (just referred to) Liponeura the strengthening of the anterior margin ("costalization" according to the terminology of ROHDENDORF) is moreover supported by sclerotization (expressed by coloring) and folding of the area between Radius and Costa. As to what extent these are already features of the ground-plan of the Blephariceridae, I don't know.
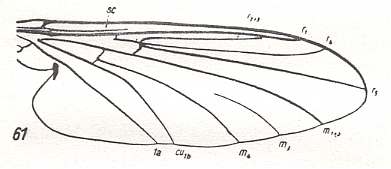
Figure 6 : Wing-venation of Bibiocephala grandis O.S. ( Blephariceridae).
(After HENNIG, 1954)
But also here there exists the tendency to reduce the number of branches of the Radial Sector : In Hapalothrix (see Figure 4 ) and Liponeura (see Figure 3, top image ) there are two of them, in Apistomyia we see only one branch, and in Hammatorrhina the Radial Sector is completely absent. See next two Figures :
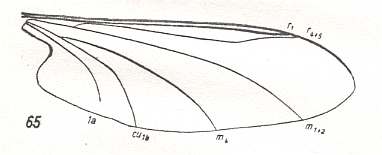
Figure 7 : Wing-venation of Apistomyia elegans BIG. ( Blephariceridae).
(After HENNIG, 1954)
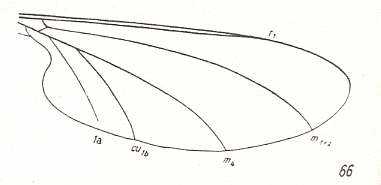
Figure 8 : Wing-venation of Hammatorrhina bella LW. ( Blephariceridae).
(After KELLOGG, 1907, from HENNIG, 1954)
In Deuterophlebiidae the [locations of the] end-points of Radius and Radial Sector (insofar as they do have end-points) are shifted toward the wing-base. Also this is a tendency we see in Blephariceridae (but much stronger still in many other diptera).
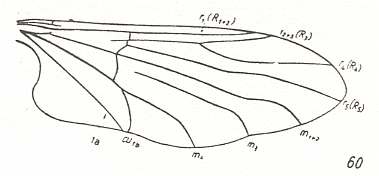
Figure 9 : Wing-venation of Paulianina hova ALEX. ( Blephariceridae).
(After ALEXANDER, 1952, from HENNIG, 1954)
In Deuterophlebiidae this shift has been carried to its extreme (see Figure 3, bottom image ). As a result of such shifts toward the wing-base all longitudinal veins are, insofar as they lie on the wing-blade (and not in the extreme wing-base), isolated. The furcation points have ended up in the basal region of the wing, where the relationship between the longitudinal veins often becomes indistinct anyway (compare for instance the Psychodidae).
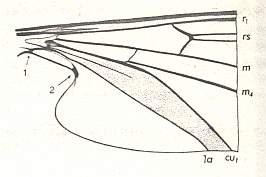
Figure 10 : Basal part of wing of Liponeura bilobata LOEW. ( Blephariceridae).
(After HENNIG, 1968)
This [sclerotized] field is absent in Deuterophlebiidae. Therefore it is perfectly possible that the 2. Anal Vein was able, thanks to the absence of the mentioned field, to extend itself a little into the wing-blade.
Formerly often the opinion was held that the similarity between Deuterophlebiidae and Blephariceridae is due to convergence as a result of the similar way of life. That may, at least for a part, be correct. However, it appears to me [HENNIG] that the similarity between them could only be so great because already the common point of departure was identical. Perhaps it is interesting to point to the Simuliidae (Black-flies) in which similar ecological conditions prevail : The pupae, being attached under water, from which the imagines [winged insects] must quickly ascend to the water-surface, where they have to rapidly unfold their wings [from the folded-up state as they are in their pupal sheaths]. [Perhaps the drying and stiffening of the wings of the emerging adult go faster in these insects as a result of the substitution of an extensive original system of genuine veins by a system of mere folds and creases (not needing to dry and stiffen)]. The wing of Simuliidae shows therefore many similarities which can be seen as adaptations to similar conditions.

However, we cannot fail to see that the similarity between the wings of Deuterophlebiidae and Blephariceridae is markedly greater than that between them and Simuliidae.
This concludes our exposition of the infraorder Deuterophlebiomorpha.
e-mail :
 ( Please write in ' Subject ' entry : ' METAPHYSICS ', in order for me to be able to distinguish your mail from spam )
( Please write in ' Subject ' entry : ' METAPHYSICS ', in order for me to be able to distinguish your mail from spam )
To continue click HERE for the further study of Organic Evolution, Part XIII.
Back to Aristotelian metaphysics Part I
Back to Aristotelian metaphysics Part II
Back to Aristotelian metaphysics Part III
Back to Aristotelian metaphysics Part IIIa
Back to Aristotelian metaphysics Part IV
Back to Aristotelian metaphysics Part V
Back to Evolutionary Part VIII