

e-mail :  ( Please write in ' Subject ' entry : ' METAPHYSICS ', in order for me to be able to distinguish your mail from spam )
( Please write in ' Subject ' entry : ' METAPHYSICS ', in order for me to be able to distinguish your mail from spam )
Preliminary examples of the type :
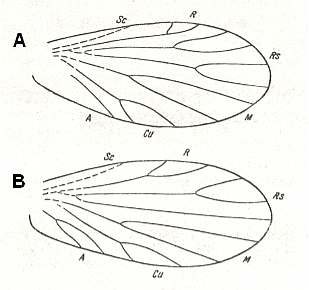
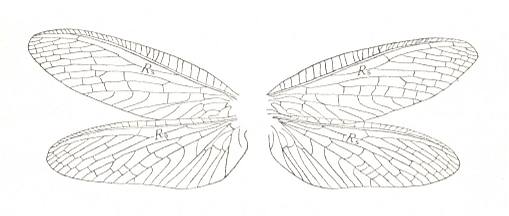
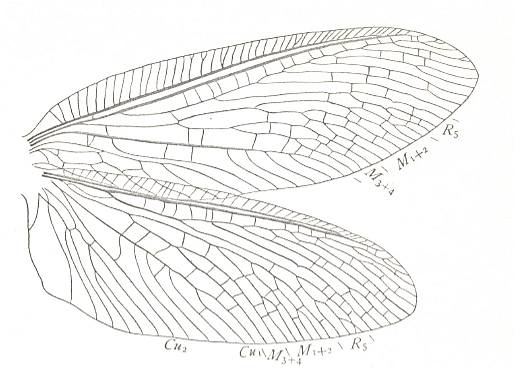
Figure 1 : Fore (A) and hind (B) wing of Archaemioptera carbonaria Guth., Miomoptera, Archaemiopteridae, upper Carboniferous of Western Europe. (After GUTHÖRL, in ROHDENDORF, 1949)
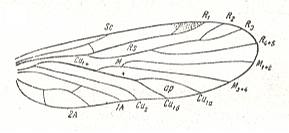
Figure 2 : Wing of Delopterum latum Sell. Miomoptera, Lower Permian of Kansas (USA). Length of (fore-)wing 6 mm, width 1.9 mm. (Carpenter, 1933).
(After TILLYARD, in ROHDENDORF, 1949)
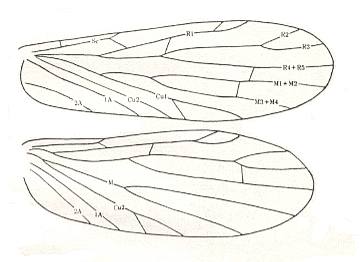
Figure 2a : Fore- and hindwings of Delopterum minutum Sell. Miomoptera, Lower Permian of Kansas (USA).
Species D. minutum : Length of forewing 4.3-4.6 mm, width 1.4 mm. Length of hindwing 4.3 mm, width 1.8 mm.
(After CARPENTER, 1933)
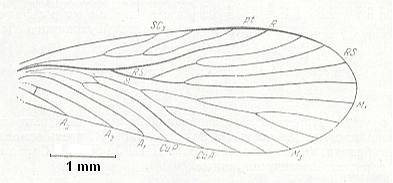
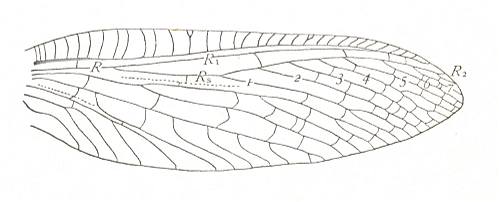
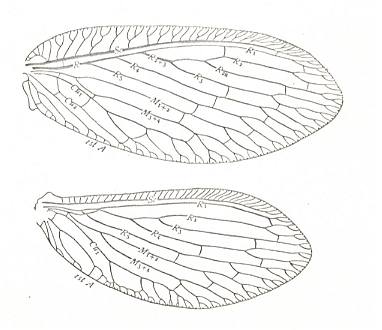
Figure 3 : Wing of Petromantis oreshnikovae O. Mart., Permochoristidae, Mecoptera. Forewing. , Permian, Basin of Kuznetz, Siberia. (After ROHDENDORF et al, 1961)
Description of the type
Morphological features
Both pairs of wings homonomous [= forewings similar to hindwings, especially with respect to size and shape], only a little different from one another, as are also the skeletons of the middle and hind thoracic segments. Muscular apparatus of the middle and hind thorax having similar structure, containing many direct and indirect muscles. Wings insignificantly elongate, of oval shape. Body elongate, devoid of aerodynamic adaptations. The wings have a moderate straigthened anterior edge and a convex posterior edge. Wing-apex often blunt. The venation is spread out more or less evenly over the whole wing-blade. Costalization little expressed. Pectinate configuration of the branches of RS [= radial sector] and M usually absent. The body is devoid of any special aerodynamic adaptations. Absolute size of the insects insignificant and varies between 5 and 12 mm. The wings are about as long as the body.
Functional features of the type
These are, as subject in their own right, almost not studied. In flight both pairs of wings move synchronously. But in this almost not showing any sign of adaptational structures fixing both wings together. Precise data on speed of flight, wing-beat frequency, load, governability, and duration of flight of the representatives of this type are unknown to me. According to qualitative observations, insects of this type fly slowly and with no notable governability. The known representatives of the neuropterygian type take flight only in certain conditions, and, possessing well developed legs, can run fast. Biologically all these insects are closely connected with the substrates in which their development takes place -- water-basins and cover of vegetation, never actively going far away from these habitats.
Differentiation and relationships of the type
Neuropterygia is with a whole series of features the most primitive wing-type, undoubtedly having been the source of the formation of the majority of the other more specialized types. And, among the examples of neuropterygia living today and having lived in the past we find various transformations in this type, bearing witness to the continuous process of the formation of its various derivatives. Most common are cases of acquiring by representatives of neuropterygia traits of long-wingedness (dolichopterygia), which consist of the elongation of the wings and body and increase of absolute size. Another kind of specialization in neuropterygia consists in the narrowing of the wing-base, which is characteristic of the type of paddle-wingedness (kopepterygia). Connections of neuropterygia with these two types are most clear. In fact, among today's insects examples of original purely expressed neuropterygia are almost absent, of neuropterygia that is, in which we do not already find traits of other, more specialized types.
As already indicated, neuropterygia was the source of formation of a whole series of other types [see Figure 6 of previous document]. Improvement of neuropterygia took place in two ways -- either the original homonomy of both pairs of wings was kept intact, or the forewings acquired other functions as compared to the hindwings. In preservation of the homonomy of both pairs [fore- and hind wings remaining similar to each other] improvement and change of aerodynamic qualities of the wings took place by a change of size of their surface-area and of their firmness. Elongation and increase of size determined the transformation of neuropterygia into dolichopterygia -- long-wingedness (in Neuroptera, Odonata). Broadening of both wings, the transformation of them into characteristic large triangular blades, created the type of platypterygia -- broad-wingedness (in Lepidoptera and certain Neuroptera). Change of firmness took place in different ways : elongated wings, -- having acquired certain traits of long-wingedness, by way of significant strengthening of their proximal parts [parts of the wing-base] and the narrowing of these parts, -- acquired characteristic traits of the type of paddle-wingedness (kopepterygia) (Odonata-Zygoptera [damselflies], Mecoptera-Bittacidae) [again, see Figure 6 of previous document, and the documents treating of the types mentioned]. Decrease of size of the insects, the working-out of passive flight [areal plankton], determined the absence of necessity to increase firmness of the wings, and the demand of their lightening [becoming organs of very low weight]. In this way the type of feather-wingedness (ptilopterygia) of the thrips (Thysanoptera) was formed.
Another course of the working-out of new types from neuropterygia was realized by the destruction of the homonomy of the fore- and hindwings [i.e. they become dissimilar to each other]. Increase of power of the wing-apparatus, increase of wing-beat frequency, determined the excessive development of the anterior pair and the transformation of the hindwings into mere appendages of the forewings, or even their complete reduction -- This was the course of the formation of the dipterygia (two-fold wingedness) of the Diptera. Finally, the acquisition by the forewings of a shielding-function took place, their stiffening and even their transformation into elytra. And with this, the hindwings broadened -- These are the courses of the formation of the various cases of straight-wingedness (orthopterygia) (Orthoptera-saltatoria) and of special forms of shield-wingedness (elytropterygia) (Sphaeropsocidae of Psocoptera).
Interrelationships of neuropterygia with other types are given in the next Figure.
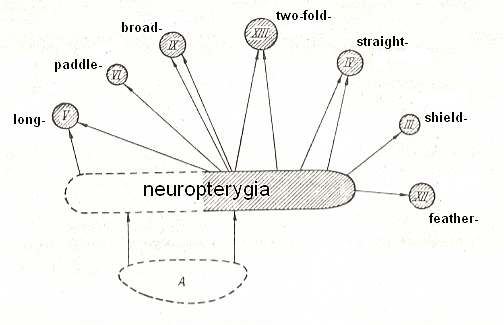
Figure 4 : Interrelationships of neuropterygia with other types.
A - hypothetical original form of flight-features (original paranotalia).
III - shield-wingedness (elytropterygia).
IV - straight-wingedness (orthopterygia).
V - long-wingedness (dolichopterygia).
VI - paddle-wingedness (kopepterygia).
IX - broad-wingedness (platypterygia).
XII - feather-wingedness (ptilopterygia).
XIII - twofold-wingedness (dipterygia).
Non-striped parts signify extinct, unknown, forms.
(Adapted from ROHDENDORF, 1949)
Representatives of the type
In neuropterygian wings Rohdendorf (1949) does not distinguish subtypes. This is because the many other types which have been derived from neuropterygia are themselves in fact its subtypes, which means that if we consider transformations and differentiation within neuropterygia, and then distinguish subtypes in it, we in fact get part of the grouping of types, chief types that is, that are indeed treated of in the following documents (long-wingedness, broad-wingedness, straight-wingedness, two-wingedness). There is, however, one special derivative of neuropterygia which as such is neither distinguished as a separate type nor as a subtype by Rohdendorf, although it deserves to be so distinguished. This derivative includes the wings of the majority of the Suborder Planipennia of the Order Neuroptera. And we will call this derivative "euneuropterygia". If we consider the latter as a subtype of neuropterygia, then it is perhaps convenient to call the remaining neuropterygian wings "proneuropterygian". We will not too explicitly take these latter to be a subtype of neuropterygia because the other subtype, euneuropterygia, might well be a chief type of its own, leaving the neuropterygian wings without subtypes.
Most typically expressed is neuropterygia in upper paleozoic Miomoptera, the wings of which show the least traits of specialization, see Figure 1, 2 and also next Figure.
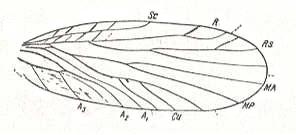
Figure 5 : Wing of Palaeomantis schmidti Handl., Miomoptera. Permian, Ural. (After MARTYNOV, in ROHDENDORF, 1949)
Fairly clearly, neuropterygia is expressed in permian Psocoptera, Homoptera (Archescytinidae, see next three Figures),
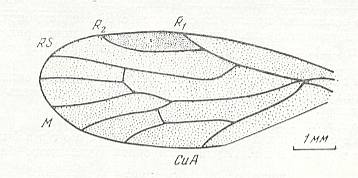
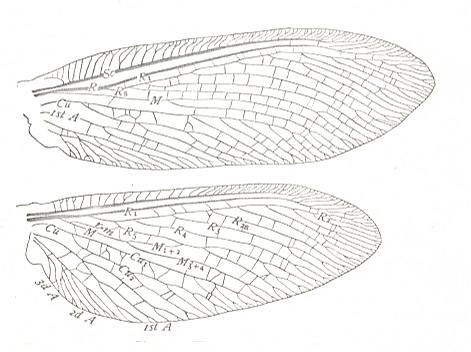
Figure 6 : Forewing of Archescytina kondomensis B.-M., Homoptera, Archescytinidae. Upper Lower-Permian, Basin of Kuznetz, Kaltan, Siberia. The wings, especially also the forewings, in the Archescytinidae are membranous. The wing-pairs are homonomous.
(After ROHDENDORF et al., 1961)
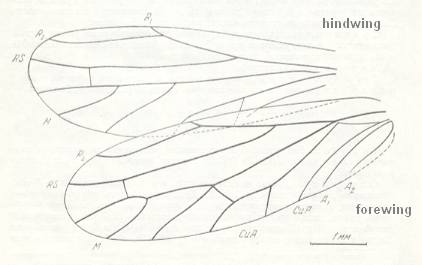
Figure 6a : Kaltanoscytina pospelovi B.-M., Homoptera, Archescytinidae. Upper Lower-Permian, Basin of Kuznetz, Kaltan, Siberia. Fore- and hindwing. Length of forewing 5.9 mm, width 2 mm. Length of hindwing 5 mm, width 1.8 mm.
(After ROHDENDORF et al., 1961)
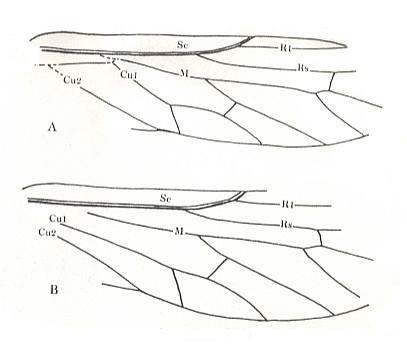
Figure 6b : Permoscytina kansasensis Carp. Family Archescytinidae, Order Homoptera. Lower Permian of Kansas (USA). Drawing of fore- and hindwing. Specimen No. 3881ab. Length of tegmen of species 11-12 mm, width 3.5 mm. (After CARPENTER, 1938)
and especially Mecoptera [scorpionflies] (Permochoristidae, Figure 3 ). Of recent insects, to this type, undoubtedly, do belong primitive Lepidoptera (Palaeosetidae, Prototheoridae, some Hepialidae), some Trichoptera (Rhyacophilidae, see next Figure
Figure 7 : Venation of Rhyacophila fuscula, Trichoptera, Rhyacophilidae. Recent.
(After COMSTOCK, with legend slightly altered, in IMMS' General Textbook of Entomology, 1977)
Beraeidae, Arctopsychidae). Here should also belong various Neuroptera ( Ithonididae, Hemerobiidae, Sisyridae, see next two Figures
Figure 8 : Right wings of Hemerobius, Neuroptera-Planipennia, Hemerobiidae. Recent.
(After RICHARDS and DAVIES in IMMS' General Textbook of Entomology, 1977)
Figure 9 : Right wings of Sisyra flavicornis, Neuroptera-Planipennia, Sisyridae. Recent.
(After COMSTOCK, 1918, in IMMS' General Textbook of Entomology, 1977)
Dilaridae), Raphidioptera (snake flies), Megaloptera (alder flies), see next Figure
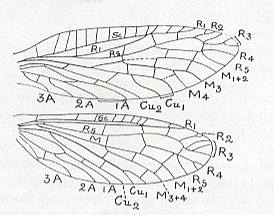
Figure 9a : Right wings of Sialis lutaria, Neuroptera-Megaloptera, Sialidae. Recent.
(After RICHARDS and DAVIES in IMMS' General Textbook of Entomology, 1977)
and Mecoptera (Panorpidae) See next Figure.
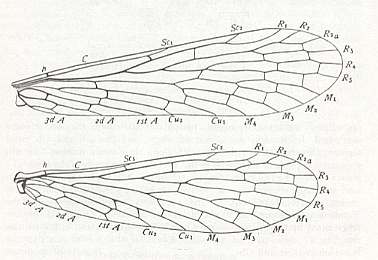
Figure 10 : Wings of Panorpa, Mecoptera, Panorpidae. Recent.
(After COMSTOCK, 1918, in IMMS' General Textbook of Entomology, 1977)
As regards these latter Orders [ Neuroptera(-planipennia), Raphidioptera, (Neuroptera-)Megaloptera, and Mecoptera], it is necessary to qualify : The structure of their wings markedly deviates from the original type characteristic of neuropterygia, namely as a result of a more elongated shape or of a higher development (secondarily) of the venation. All these forms, undoubtedly, have already survived the phase of the most simple form of neuropterygia and are under way in working out new types -- long-wingedness and others (see Figure 4 ).
In addition to the above figured representatives of neuropterygia there are a few members of the extinct Order Megasecoptera (Carboniferous-Permian) of which the wings can be assigned to the present type, neuropterygia :
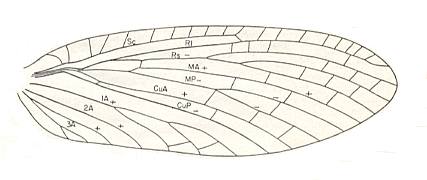
Figure 11 : Parelmoa revelata Carp. Family Elmoidae. Order Megasecoptera. Drawing of forewing. Fore wing : length 15 mm, width 5 mm. Hindwing unknown. Lower Permian of Oklahoma (USA). Of the family Elmoidae it is known that they are neopterous, i.e. that they are able to fold their wings back over or along the abdomen at rest.
(After CARPENTER, 1947)
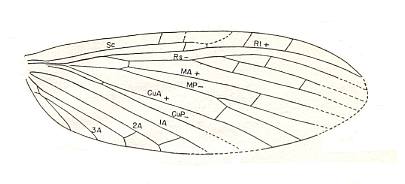
Figure 12 : Parelmoa radialis Carp. Family Elmoidae. Order Megasecoptera. Drawing of hindwing. Length 12 mm, width 4 mm. (Forewing length 15 mm, width 5 mm). Lower Permian of Oklahoma (USA).
(After CARPENTER, 1947)
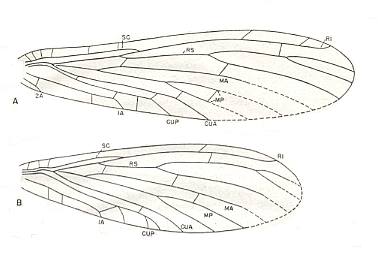
Figure 13 : Elmoa trisecta Till. Family Elmoidae. Order Megasecoptera.
A - Forewing : drawing based mainly on specimen no. 4604.
B - Hindwing : drawing based chiefly on specimen no. 4590.
The lengths of the fore- and hindwings in one and the same specimen are equal.
Forewing length 12 mm, width 3.2 mm. Hindwing length 12 mm, width 4 mm.
(After CARPENTER, 1943)

Figure 14 : Martynovia insignis Till. Family Martynoviidae. Order Megasecoptera.
A - Forewing : drawing based chiefly on specimen no. 4601.
B - Hindwing : drawing based mainly on specimen no. 4604, and the holotype.
Length of forewing of species : 12.5-20 mm, width 3-4.7 mm. Length of hindwing of species : 12.5-20 mm, width 4.2-6 mm.
Although the forewing of this species has a more or less orthopteroid look, it doesn't belong to the wing-type orthopterygia (and thus also not to its subtype neurorthopterygia), because orthopterygian wings are markedly heteronomous, whereas those of Martynovia insignis are largely homonomous.
(After CARPENTER, 1943)
The wings of Elmoidae (Megasecoptera) seem -- as to wing-type -- to remind us of those of Paraplecoptera [stonefly-like extinct insects]. But the decisive typological difference between them is that the wings of Paraplecoptera are heteronomous whereas those of Megasecoptera are homonomous or nearly so. The other members of the Order Megasecoptera have wings that are assigned to the type kopepteygia (paddle-wingedness).
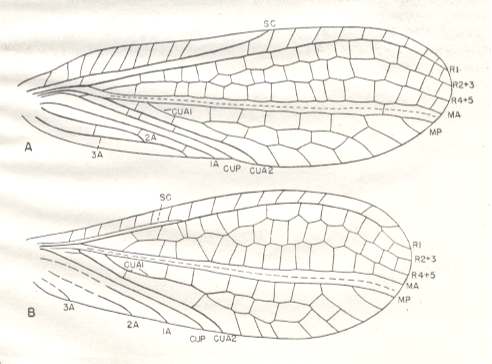
Figure 14a : Permoberotha villosa Till. Family Permoberothidae. Order Neuroptera-Planipennia.
A - Forewing : drawing based chiefly on specimens no. 4605 and the holotype.
B - Hindwing : drawing based mainly on specimen no. 4575.
Length of forewing : 9 mm, width 2.8 mm. Length of hindwing : 8 mm, width 2.8 mm.
(After CARPENTER, 1943)
The body structure of this insect is very incompletely known. The length of the entire body is 6.5 mm, a little less than that of the forewing. The abdomen is slender, 4 mm long and 1 mm wide.The abdomen terminates in a pair of cerci.
Among fossil insects there are some that, although -- as to their wing-venation -- well-preserved, cannot be assigned with certainty to any known (fossil or recent) insect order. This indicates that the ordinal diversity of insects generally is greater than that suggested by today's classification of fossil and recent insects. Among such "order unknown" insects are the Apheloneuridae, found in the lower Permian insect bed of Elmo, Kansas, USA. Of the genus Apheloneura we know both wing pairs. They are such that the wings might be assigned to the present type, neuropterygia :
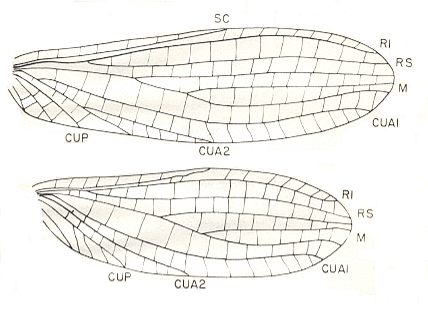
Figure 15 : Fore (top) and hind (bottom) wings of Apheloneura minutissima Carp., Family Apheloneuridae. Order unknown. Lower Permian of Kansas (USA). Length of forewing 3 mm. Width 1 mm. Length of hindwing 2.7 mm. Width 1 mm.
(After CARPENTER, 1976)
Also another species of Apheloneura is described (Carpenter, 1976), A. amplia Carp.
Euneuropterygia
Neuropterygia has given rise to many, if not most, other wing-types, including [having given rise to] the wings of all of the recent representatives of the Order Neuroptera. And the wing-venation in the latter, instead of having been subjected to all sorts of reductions, has, in many cases, undergone extensive proliferation, i.e. the venation has become richer still, as a result of many veins and veinlets having been added to the initial neuropterygian venation. And what is especially significant is the fact that in the forewings of most Neuroptera the costal area has widened, the subcosta (SC) is long, more or less remote from the anterior wing-margin, but running very close and parallel to the Radial vein (R + R1). Further, the branching of the Radial Sector (RS) has been transformed from dichotomic to pectinate. All this forms a special venational configuration which undoubtedly has some functional significance [eh, has it really?, see below], and the wings that have it surely deserve a subtype of neuropterygia of their own (if not, a type of their own). Therefore, most of the above figured representatives of the type neuropterygia could be assigned to a subtype of it that might be called Proneuropterygia, or, equivalently, primitive neuropterygia, while most of the members of the Order Neuroptera, of which the wings possess the just mentioned characteristics, can be -- as to their wings -- assigned to the subtype Euneuropterygia (eu-neuro-pterygia), or, equivalently, true neuropterygia, of the type neuropterygia.

An unidentified recent species of the Neuroptera-Planipennia, with wings typical of euneuropterygia. Length of forewing of the insect about 10 mm. Dorsal view. Specimen scanned with resolution 600 (HP PrecisionScan Pro).
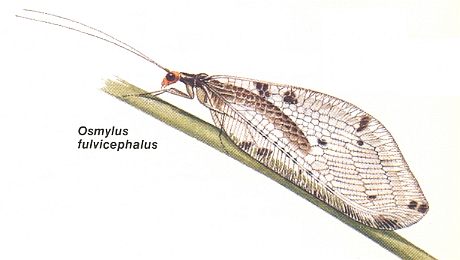
Most characteristically expressing the subtype euneuropterygia are the wings of Osmylus :
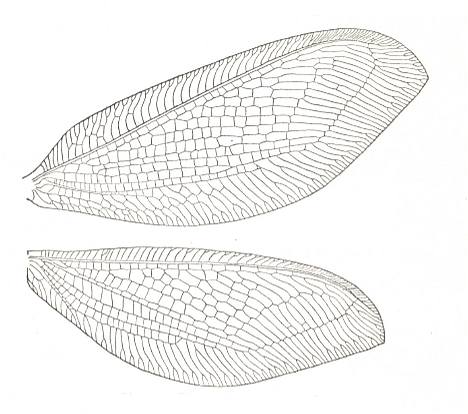

Extreme development is perhaps best illustrated by the wings of Psychopsis :
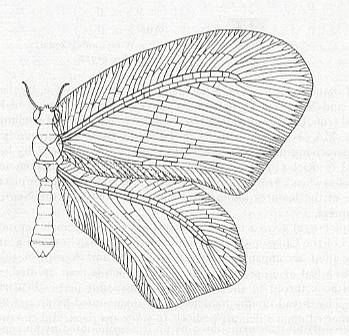
All the wings belonging to the euneuropterygian subtype exclusively belong to representatives of the Suborder Planipennia of the Order Neuroptera. Of this suborder there exist about 4500 recent species, distributed among 17 families forming five family-groups :
Before we depict some of the many other wings (than the ones already depicted) that belong to the euneuropterygian subtype, we first depict a series of wings that shows the formation of euneuropterygia from proneuropterygia, [this formation] largely being visible already within the recent representatives of the Order Neuroptera (consisting of the two suborders Megaloptera, i.e. alder flies and snake flies [Sialidae, Corydalidae, Raphidiidae] and Planipennia, i.e. lacewings, ant lions, etc. [ Ithonidae, Osmylidae, Sisyridae, Hemerobiidae, Psychopsidae, Myrmeleontidae, etc.] ), and [this formation being] further clarified by certain paleozoic fossils. Only after this series we grade into the representatives of true euneuropterygia. As regards the following Figures of wing-venation, the interpretation of the venation, expressed in the lettering of the veins in these Figures, is of the various authors who have given these Figures.
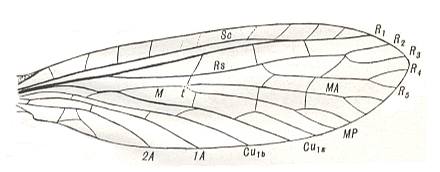
Here we see the enlarged costal area, provided with veinlets, the alignment of SC and R-R1. But still the longitudinal veins, coming after the Radius (R1) [especially the branches of RS] have not yet arranged themselves pectinately.
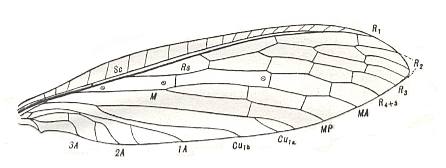
Here we see the more typically enlarged costal area, provided with a regular series of veinlets, the still closer alignment of SC and R-R1, and at least the beginnings of the arrangement of the branches of RS into a pectinate series.
Here we see the well developed costal area with a regular series of veinlets, the alignment of SC and R-R1, and the arrangement of the branches of RS into a perfect pectinate series.
As in all proneuropterygian wings the pairs are largely homonomous. The anal region of the hindwings is a bit expanded and separated from the wing's main part.
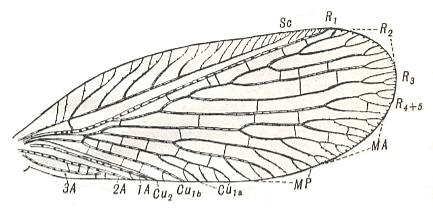
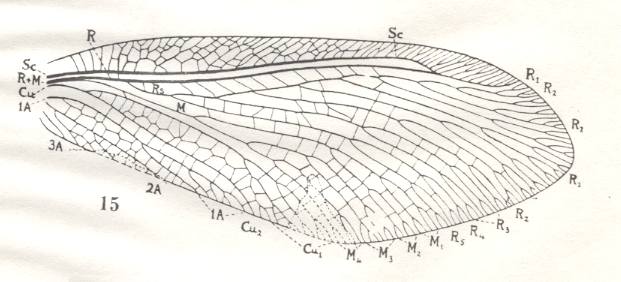
Figure 20a : Permorapisma biserialis Till. Forewing. Length 19 mm. Greatest breadth 7 mm. Family Permithonidae, Order Neuroptera-Planipennia. Upper Permian of Belmont, New South Wales, Australia. (After TILLYARD, 1926)
Here we see, figures 20 and 20a, that the euneuropterygian subtype of neuropterygia was fairly well developed already in permian times.
Also here the wing pairs are largely homonomous. The anal area of the hindwing is slightly more expanded than it is in the forewing.
Also here the wing pairs are largely homonomous. The anal area of the hindwing is not expanded. The hindwing is smaller than the forewing.
Also here the wing pairs are largely homonomous. The anal area of the hindwing is very slightly more expanded than it is in the forewing.
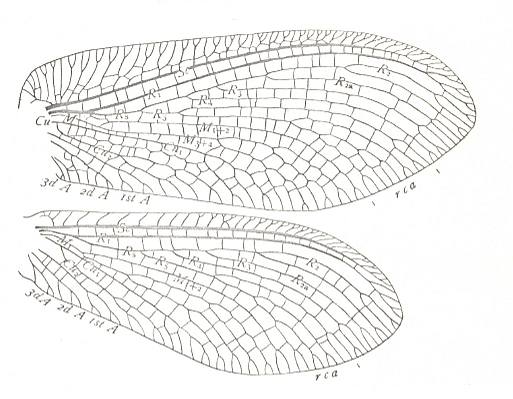
Figure 24 : Wings of Rapisma viridipennis. Probably family Ithonidae. Order Neuroptera-planipennia. Recent. rca - radial cuneate area. (After COMSTOCK, 1918)
Also here the wing pairs are largely homonomous. The anal area of the hindwing is not expanded. Costal space in forewing basally very broad.
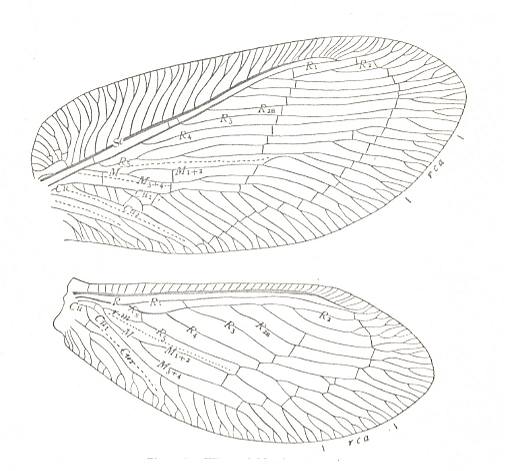
Figure 25 : Wings of Megalomus maestus. Family Hemerobiidae, Order Neuroptera-planipennia. Recent.
rca - radial cuneate area.
(After COMSTOCK, 1918)
Anal area of hindwing not expanded. Costal space in forewing very broad.
And finally, then, we have fully-fledged euneuropterygian wings in, for example, Osmylus (depicted earlier) :

Figure 26 : Wings of Osmylus hyalinatus. Family Osmylidae, Order Neuroptera-planipennia. Recent.
(After COMSTOCK, 1918)
Anal area of hindwing not expanded. Costal space in forewing broad.
A long-winged variant of euneuropterygia we see in ant lions :
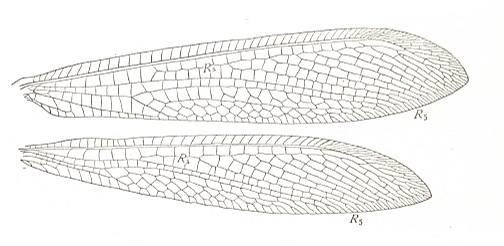
Figure 27 : Wings of Myrmeleon, Family Myrmeleonidae, Order Neuroptera-Planipennia. A species from Katihar, former British India. Recent. (After COMSTOCK, 1918.)
The family Myrmeleonidae is with about 1200 species the largest family of the Planipennia. It also contains the largest species of this Suborder. The adults look like damselflies or dragonflies (Odonata) but in resting position they hold their wings roof-like over the abdomen. Apart from this, their typical planipennian wing-venation (different from that of the Odonata) shows them to belong to the Order Neuroptera. As imago most species are twilight or nocturnal animals. During the daytime they hide or relatively quietly sit on lower vegetation, bushes and trees. Only now and then they take to a slow fluttering flight that brings them a few meters farther. Their imaginal life lasts only a few days up to a few weeks. During this time they are predatory, especially attacking insects, thereby taking in relatively not much food. The larvae are also predatory, a small part of the species constructing the well-known funnels to catch small arthropods.
The wings of Myrmeleon show the transition from the type of neuropterygia to the type of dolichopterygia (long-wingedness). And indeed we also have placed these wing there. But we must say that they 'project' as it were the euneuropterygian subtype of neuropterygia into the type dolichopterygia.
But neuropterygian wings can also give rise to platypterygia (broad-wingedness) :
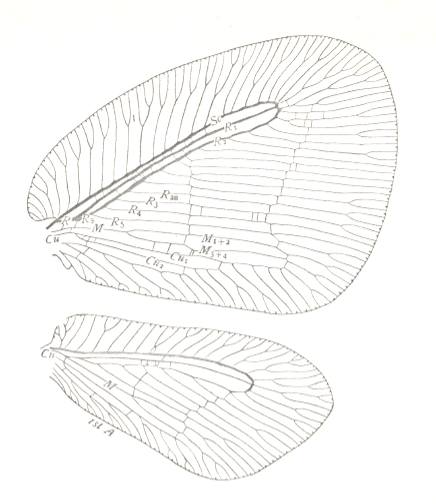
Hindwing much smaller than forewing. Shape of hindwing differing from that of forewing, but not as hindwings usually do in insects. See also figure above.
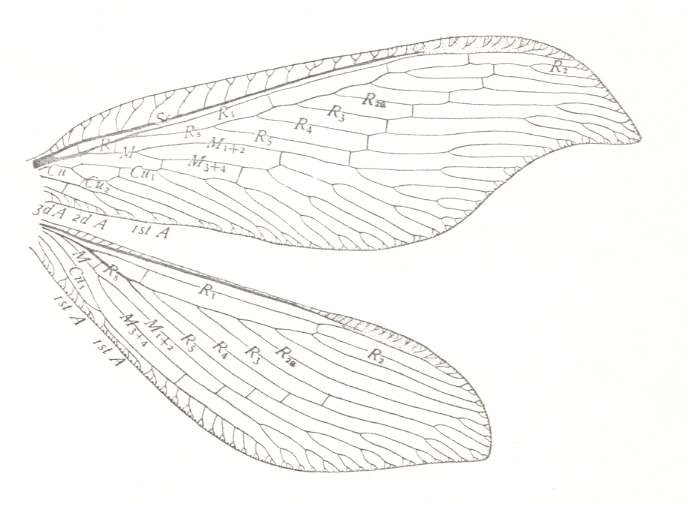
Figure 29 : Wings of Berotha insolita, Family Berothidae, Order Neuroptera-Planipennia. Recent. (After COMSTOCK, 1918.)
Shape of forewing specialized. Basal part of hindwing narrower than that of forewing. Wing pair far removed from any trace of the, we might say, orthopteroid configuration in which the basal part of the hindwing is always broader than that of the forewing, and the anal (or ano-jugal) area well developed and contrasting with the rest of the hindwing, and, finally, in which [orthopteroid configuration] the forewing usually is more or less elongate with subparallel margins.
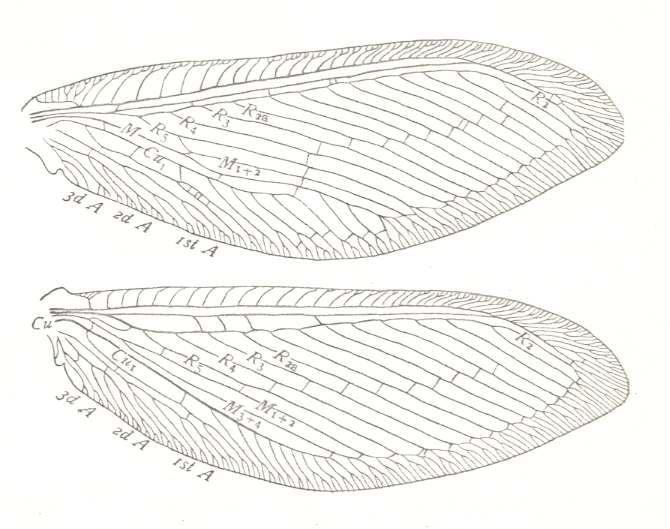
Figure 30 : Wings of Polystoechotes punctatus, Family Polystoechotidae, Order Neuroptera-Planipennia. Recent.
(After COMSTOCK, 1918.)
Wing pairs perfectly homonomous. Seam of terminal twigs around the wing just as well developed in hindwing as in the forewing.
Yet some other examples of the extraordinary evolutionary plasticity of euneuropterygian wings are given here. In fact wings of yet another different type are created, namely those of the type uropterygia, or, equivalently, tail-wingedness :
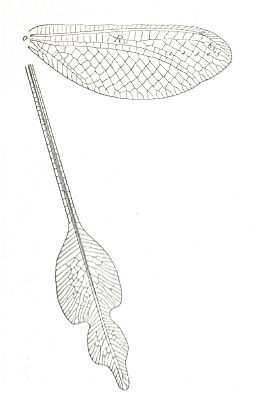
Figure 31 : Wings of Oliverina extensa, Family Nemopteridae, Order Neuroptera-Planipennia. Recent. o - oblique vein, vein M3+4.
(After COMSTOCK, 1918.)
Wing pairs hyper-heteronomous. In the forewing the euneuropterygian type of venation is still retained : Broad costal field with regular veinlets extending from Subsosta to Costa. Subcosta and Radius parallel. Radial Sector pectinately branched. But of course the extreme heteronomy of the wings places them outside the euneuropterygian subtype, and even altogether outside the neuropterygian type, and places them into the type uropterygia.
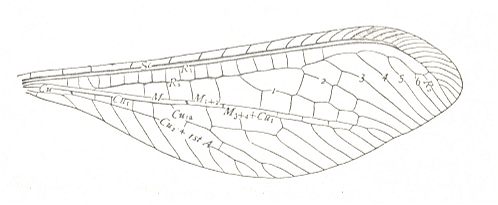
Figure 32 : Forewing of Croce filipennis, Family Nemopteridae, Order Neuroptera-Planipennia. Recent. Wing-type : uropterygia (hindwing ribbon-like). (After COMSTOCK, 1918.)
Also here the euneuropterygian type of venation is more or less preserved in the forewing.
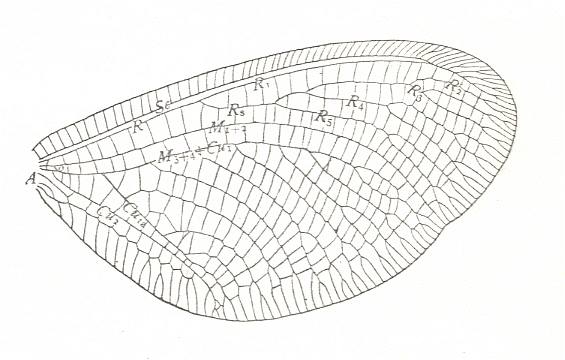
Figure 33 : Forewing of Nemoptera sinuata, Family Nemopteridae, Order Neuroptera-Planipennia. Recent. o - oblique vein, vein M3+4. Wing-type : uropterygia (hindwing ribbon-like). (After COMSTOCK, 1918.)
Here the euneuropterygian type of venation is well preserved in the forewing.
From all this it is clear that the Neuroptera-Planipennia (i.e. the Neuroptera proper), the representatives of which have roughly the same way of life, followed an evolution that was determined by the diversification of their wings. This diversification not only has resulted in a great variability within the euneuropterygian subtype, but also in the origin of a number of other (main) types such as platypterygia (broad-wingedness), dolichopterygia (long-wingedness), and uropterygia (tail-wingedness). If the wings in the Order Neuroptera-Planipennia were considered to represent one single type of their own, all the mentioned types derived from euneuropterygia were direct subtypes of the Neuroptera-planipennian type. And when considering the representatives of all three Suborders of the Order Neuroptera (Megaloptera, Raphidioptera, and Planipennia) to represent one single wing-type, then the number of possible subtypes of it would increase still more. Indeed, further below, all the representatives of the Order Diptera will be considered to represent a single wing-type, in fact a single subtype. It is the subtype eudipterygia of the type dipterygia. And in Part V of Website we have divided the wings of the Order Diptera into a number of subtypes (there called "types") and subtypes of them.
General theoretical remarks about euneuropterygian wings.
The representatives of the subtype euneuropterygia, with their elaborate many-veined roughly homonomous wings, make us think more deeply about the essence and significance of the venational pattern in, and general structure of, insect wings.
It turns out that each insect Order, or Suborder for that matter, has its own typical wing structure, especially its own typical wing-venation. Indeed, the representatives of each such Orders have wings that possess some basic general scheme of venation from which more or less minor deviations may take place during evolution, but always, apparently, within certain limits set by the qualitative basic content (basic set of features) of the particular insect Order (or Suborder). And although the gross features of such an Order-specific general scheme of venation and also the general structure of the wings (homonomy, shield properties, wing-folding adaptations, etc.) may have in most cases a functional significance (which is the main criterion of Rohdendorf's types and subtypes), some of these general schemes are not at all functional. This is, for example, the case in the euneuropterygian wings of most of the Neuroptera-Planipennia. But the same is true of the wings in the Order Hymenoptera (wasps, bees, and ants). In the latter only the system consisting of a more or less elongate costalized forewing physically connected (by hooks) to a small hindwing with only a few veins, is a truly functional system, as such expressing a subtype of dipterygia. But yet the general venation in Hymenoptera, although originally consisting -- like in any other winged insect -- of (1) the Costal system, (2) the Subcostal system, (3) the Radial system, (4) the Medial system, (5) the Cubital system, the (6) Anal system, and, finally, (7) the Jugal system (the latter often having disappeared), is highly typical : hymenopterous wings can directly and without doubt be recognized as such. And precisely this typical look of their venation does not have, as it seems to me, any functional significance. The same is entirely true of the wings of the Planipennia, while other insect Orders may have, on the other hand, a general wing structure that evidently does have such a significance, like the wing apparatus of beetles (Order Coleoptera), where the forewings have acquired a shielding function, while the flight function has come to lie solely in the aerodynamically worked-out hindwings. Also in Orthoptera (grasshoppers, locusts) the general wing structure has functional significance : The elongate forewings are functionally the anterior part of the broadened hingwings, while in other Orthoptera such as crickets the forewing has, apart from a shielding function, a sound-producing function. Further the long wings of dragon-flies function as powerful flight-organs, and the rich venation serves the stiffening of these wings. Further, the broad wings of butterflies enable a flight of the gliding type, and so on. Nevertheless, if we descend further into details, also these Orders (Orthoptera, dragon-flies, butterflies, etc.) have a typical wing-venation of their own. And although often this (further analyzed) wing-venation has some functional significance, for example determining certain details of the flight-regime of the insect, the very fact of its evident morphological typicality (typical look of the venation, the condition of the main-vein systems, characteristic of a given insect Order or Suborder), its Order-specific unique structure, has no functional significance. And this is especially evident in the wings of the Planipennia : The representatives of the latter are weak and not persistent flyers, their forewings lack special shielding features (such as partial hardening or stiffening), they lack sound-producing structures, and both wing pairs lack features of camouflage or deterrence. Yet the venation is morphologically not at all indifferent, it is not regressive, but on the contrary, it is highly elaborated with a complex and persistent venational pattern.
How can such an elaborate venational pattern be functionless?
The answer to this question is difficult indeed. Let us try. Because the general venational scheme is functionless it may express in a codified way the basic qualitative content, 'whatness' or identity of the Order or Suborder to which its representatives belong. In our theory of the Implicate and Explicate Orders (our noëtic theory of evolution), developed in Fifth Part of Website, we have theorized that the evolution of organisms does take place, not in the Explicate Order, i.e. not in the physical material order of reality, but in the immaterial Implicate Order. In that Order strategies for immaterial patterns to materially exist in the Explicate Order are being worked out, because every immaterial pattern intrinsically aspires to become ontologically complete, i.e. to become material. But a material pattern can only exist as such when it complies with appropriate existential conditions. Many immaterial patterns, can, without any further working-out of them, exist materially in the Explicate Order, and be there in the form of, for instance crystal lattices. But there are many immaterial patterns residing in the Implicate Order that cannot exist in the Explicate Order without any further qualitative additions and changes (apart from matter). So for them to be able to exist in the Explicate Order (and then having become ontologically complete) they have to become s t r a t e g i e s, strategies, that is, to exist and persist in the Explicate Order. And when they then appear as materialized strategies in the Explicate Order (i.e. when they 'project' into that Order) they appear there in the form of what we are used to call organisms. Indeed, organisms are materialized strategies having immaterially developed in the Implicate Order. And this development in the Implicate Order is a timeless 'process' that we may best compare with mathematical derivations of one immaterial pattern from another according to certain rules inherent in the Implicate Order. Now the sequence of actual physical appearance in the Explicate Order of these strategies, of these organisms, is what we call evolution. And the consecutivity of the mentioned appearance (order of projections) depends on two factors : firstly on the consecutivity of formal derivation of one immaterial pattern of strategy from another (in the Implicate Order), and secondly of the actual presence in the Explicate Order of the appropriate external existential conditions, i.e. the presence of vacant ecological niches. We can say that the actual organisms existing in the Explicate Order constitute the m e a n i n g of the corresponding immaterial patterns of strategy developed (and present) in the Implicate Order, i.e. any such mentioned immaterial pattern is a message, while the corresponding material organismic species represents the meaning of that message. And, of course, also in insects every insect species, existing or having existed in the Explicate Order, represents the physical meaning of an immaterial message developed in the Implicate Order. Now because the wing-venation in any species of Pterygota (winged insects), insofar as it is basic and typical of the insect Order of which that species is a representative, is not determined by functional factors, and because it does not interfere with the rest of the insect body (it is all by itself), it may in turn represent a formal expression of the qualitative content, the identity or whatness, of that Order, but then only of the very formal content of that Order. All this largely complies with the actual situation, practise and experience in the classification of insect Orders and Suborders. And even when the wing-venation turns yet to be somehow functional in its nature (i.e. functionally determined), the t y p i c a l structure which makes this function possible is as such, i.e. as being typical or characteristic, functionless, because the same function may be made possible by other structures as well. This is well illustrated by a comparison of the representatives of the family Mantispidae (Neuroptera-Planipennia) and those of the Order Mantodea (praying mantises). Both have the same way of life, ambushing insects, and perform it with the same tools, having their forelegs transformed into strong snapping devices. Also the shape of the body and head is almost the same in both forms. But the general structure of the flying apparatus and especially its venation definitely shows the one to belong to the Order Neuroptera and the other to the Order Mantodea. See next five Figures :
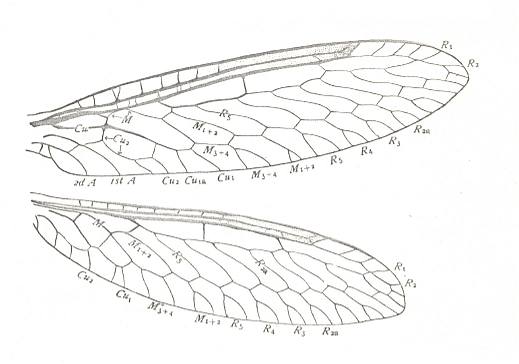
Figure 34 : Wings of Climaciella brunnea, Family Mantispidae, Order Neuroptera-Planipennia. Recent.
(After COMSTOCK, 1918.)
Wing pairs homonomous, hindwing only a little shorter than forewing. Venation and shape similar in both wings.
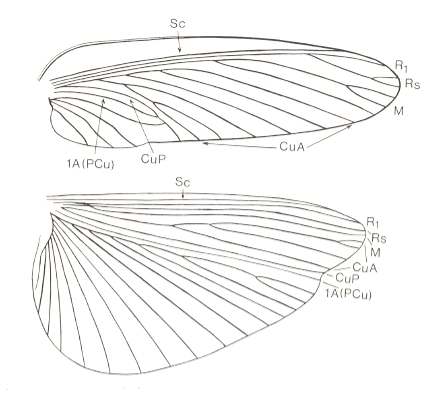
Figure 35 : Wings of Sphodromantis viridis, Order Mantodea. Recent.
(After RAGGE, 1955, in HENNIG, 1969)
A typical orthopteroid wing apparatus. Anal area of hindwing strongly expanded, distinguishing itself from the rest of the wing [ I don't know whether cross-veins are absent or omitted, therefore I give a second Figure of wings of Mantodea] :
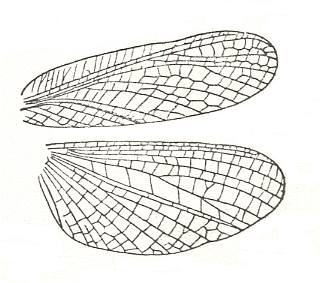
Figure 36 : Wings of Gonypeta sp., Order Mantodea. Recent. Length 14 mm.
(After HANDLIRSCH, in ROHDENDORF, 1949)
Also a typical orthopteroid wing apparatus. Anal area of hindwing moderately expanded, but more or less clearly distinguished from the rest of the wing.
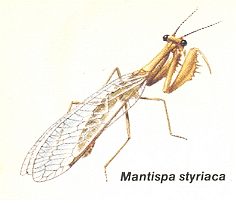
Figure 37 : Mantispa styriaca. Family Mantispidae, Order Neuroptera-Planipennia. Recent. Length about 18 mm.
(After CHINERY, in Nieuwe Insektengids, 1987)

Figure 38 : Perlamantis alliberti. Family Amorphoscelididae, Order Mantodea. Recent. Length about 18 mm.
(After CHINERY, in Nieuwe Insektengids, 1987)
So it is well founded when we suppose that the kernel of the qualitative content [set of properties] of some larger taxonomic unit, say a family, suborder or order, of winged insects, is, like a fingerprint, spatially and topographically expressed in the structure, venation, form and shape of the wings that are dominant in that taxonomic unit. Of course, this typical wing-structure, expressing the kernel of the given taxonomic unit, may get lost under the pressure of certian functional demands. So in the wings of the representatives of the family Coniopterygidae, agreed to belong to the Neuroptera-Planipennia, nothing is left of typical planipennian wing-venation, and even the homonomy of the wing pairs is affected :
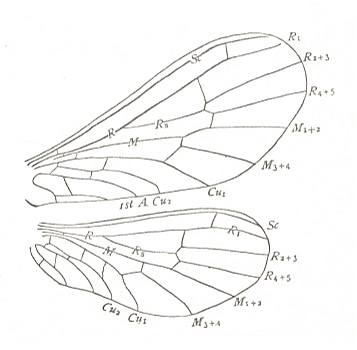
Figure 39 : Wings of Semidalis aleurodiformis, Family Coniopterygidae, Order Neuroptera-Planipennia. Recent.
(After COMSTOCK, 1918.)
Wing pairs (moderately) heteronomous, but venation still similar in both wings.
Representatives of Coniopterygidae : Body length, 2-4 mm. Wingspan 5-8 mm. They are the smallest Planipennia.
Surveying the wings of the Planipennia -- we, here, have depicted only a small part of their diversity -- shows us a given definite venational configuration that is common to, or dominant in, all of them (except in Coniopterygidae). All differences consist in more or less small variations on this common scheme. Generally this is a well known phenomenon throughout all insects : some one definite persistent common or dominant venational configuration from which small variations depart into all directions. We see it in the wings of the Order Hymenoptera, in those of the Suborder Anisoptera of the Order Odonata, in the family Syrphidae (hoverflies) of the Order Diptera, in the family Psychodidae (moth-flies) of the Diptera, in the Order Lepidoptera, etc., to cite just some random examples. A still deeper generality of wing-venation consists in the fact that the wings of all insects have a venation that derives from a scheme of seven vein-systems : (1) the Costal, (2) Subcostal, (3) Radial, (4) Medial, (5) Cubital, (6) Anal, and (7) Jugal system.
We may theorize (in the sense of "if it were indeed the case, then . . . " ) that most of the non-functional structures of any winged insect are located in the wing-venation, because the latter least interferes with other structures of the insect. But these non-functional elements can only exist (in the wings) when they do not interfere with the overall functional structure of the wings which surely are organs of flight. Indeed, the wing-types described by Rohdendorf are supposed to be functional types, realizing particular ways of flight (such as passive, gliding, or traction flight). And as regards the wing-venation itself, costalization (not only consisting of a crowding and strengthening of anterior veins, but at the same time of a lightening of the posterior part of the wing) is certainly in many cases a functional characteristic, enabling high wing-beat frequencies. But apart from these evident functional demands imposed on the structure of the wings as organs of flight, we may further theorize that the wing-venation in the majority of winged insects is functionless (this is especially true of more or less stiffened forewings -- tegmina). If so, the venational scheme in the wings of the representatives of most species of Pterygota expresses in some way the (qualitative content of the) original immaterial form that has, in the Implicate Order, developed into the strategy-form which, after projection into the Explicate Order, gives rise to such a species (i.e. the single immaterial strategy-form becomes expressed in the Explicate Order in the form of material individuals). When there are, in the Implicate Order, say, several similar (but not identical) original immaterial forms, they will not necessarily develop into similar (among each other) strategy-forms, but may result in quite different (from each other) strategy-forms (divergence), in the Explicate Order then involving different material structures in the species concerned. Or, the other way around, dissimilar original immaterial forms may develop into quite similar (among each other) strategies (convergence). So if we would classify insects, basing ourselves solely on their noëtic descent from original immaterial forms, i.e. if we would classify these forms themselves (with the help of non-functional characters in the materialized strategies), the result would be quite different from that of current classification (phylogenetic or typologic), because in the latter no distinction is made between functional and non-functional characters. Current classification is indeed a classification of fully-fledged strategies, i.e. of organismic species, as to what they themselves precisely are and how they connect with other species. As such this classification is meaningful and we are not intended to replace it. We only want to place it into a broader theoretical context, which is in fact a broader ontological context (a context of Reality and Being). Every original immaterial form in the Implicate Order, - when it is such that it cannot, as it is by itself, be directly projected into the Explicate Order and so acquire ontological completion, - will, 'subsequently', develop into a strategy-form that can be so projected, without, during this development, losing its original identity. In this way each such original immaterial form gives rise to a certain definite organismic species, and this independently of other such original immaterial forms. In the Implicate Order this results in a great many parallel lines of development, giving, after projection, rise to independent organismic species.
This is then the overall picture we assume of the polyphyletic development of organisms.
And so the situation as found in the Neuroptera-Planipennia has inspired us to this extension of our noëtic theory of evolution.
With this we have concluded the exposition of neuropterygia. In the following document we will describe the next wing-type, blattopterygia.
e-mail :
 ( Please write in ' Subject ' entry : ' METAPHYSICS ', in order for me to be able to distinguish your mail from spam )
( Please write in ' Subject ' entry : ' METAPHYSICS ', in order for me to be able to distinguish your mail from spam )
To continue click HERE for further study of the types of flight-devices in insects, Part III, Blattopterygia.